A discovery presented by Catherine Seva, biologist at Inserm
Two research scientists, Catherine Seva and Audrey Ferrand, from a combined team of researchers from Inserm / Université Toulouse III – Paul Sabatier (Cancerology research center of Toulouse), have just developed a test to predict the risk of colorectal cancer. This test, based on the presence of a protein known as progastrine, can predict the risk of tumors occurring in patients who have already undergone surgery to remove polyps that were considered to be benign. In practice, these tests could be used for routine screening on resected polyps in order to identify patients presenting a high risk of developing precancerous lesions and for whom no monitoring is recommended at present. The results of this work are published in the review Cancer prevention research.
Bowel cancer is the 4th highest cause of death from cancer in France. So March 2012 has been named “Blue March”, colorectal cancer awareness month.
A predictive test to assess the risks of bowel cancer
Colorectal (or bowel) cancer is the second most frequent cancer. Hyperplasic polyps are the most frequently occurring colorectal lesions Nearly one quarter of the European population between the ages of 20 and 54 will develop this type of polyp. For a long time, these lesions were considered to be benign, so currently there is no recommended monitoring of these patients once the polyps have been removed surgically. However, some of these polyps could be precursory signs of bowel cancers. Up until now, we had no way of identifying the sub-group of polyps with potential for malignancy.
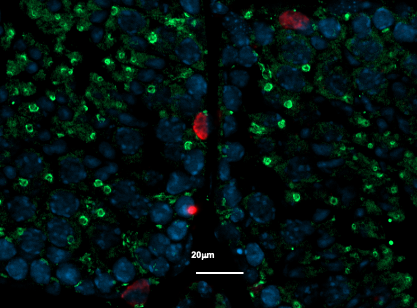
So with the aim of finding a biomarker that could predict the risk of bowel cancer in patients presenting with hyperplasic polyps – the most benign type – Catherine Seva and her co-workers carried out a 10-year retrospective clinical study. They analyzed the presence of a particular protein name progastrine in hyperplasic polyps from 74 patients. Progastrine is already known to play a part in colic cancerogenesis. This protein, produced by tumerous colorectal cells, is not present in healthy bowel cells. So the researchers tried to find out whether the expression of this protein could predict the occurrence of cancerous lesions in the years following surgical resection of the polyps. “When we first put forward this hypothesis, we had in mind the idea that this was a step towards ensuring suitable monitoring and very early detection of colorectal cancer”, explains Catherine Seva, Research Director at Inserm.
Thanks to these analyses, the research scientists showed that there was indeed a significant relationship between high levels of progastrine and future occurrence of precancerous lesions. And yet although these polyps had been considered as benign and risk-free, within a period of 2 to 10 years, 100% of patients who presented high levels of progastrine developed adenomas, renowned as early colorectal cancer lesions. Inversely, in patients who expressed little or no progastrine, no lesion developed at all over the 10 years following the removal of their polyps.
Based on these results, the researchers compiled a predictive test based on the age of the patient and prograstrine immunohistochemical marking. This highly sensitive and specific tests can predict tumors in patients who have developed hyperplasic polyps.
“There is no recommended monitoring at the present time, and yet measuring the prograstrine expression in hyperplasic polyps could identify the population of patients who have a high risk of developing a precancerous lesion”, concludes Audrey Ferrand, research scientists at Inserm and signatory of this paper.
As a result of this study, it could be envisaged to include a larger number of patients to valid this as a routine test.
This research has been patent-protected by Inserm Transfert.