Researcher Contact
Christine Baldeschi
Unité Inserm 861 “Institut des cellules souches pour le traitement et l’étude des maladies monogéniques (I-STEM)”
+ 33 (0) 1 69 90 85 33
rf.metsi@ihcsedlabc
Birthmarks, café au lait spots—although usually harmless (over 10% of the population has one or two), the presence of more than 5 of them in a child can conceal a hereditary disease, neurofibromatosis type 1 (NF1).
This is one of the most common genetic diseases (affecting 1 in 3,000 individuals). Until now, the molecular mechanisms associated with altered skin pigmentation were poorly understood, particularly since few appropriate models have been developed in the laboratory.
Inserm researchers at I-Stem (Institute for Stem Cell Therapy and Exploration of Monogenic diseases) have developed a model reproducing in vitro the hyperpigmentation associated with type 1 neurofibromatosis from embryonic stem cells carrying the mutation responsible for this disease[1].
By establishing a protocol for differentiating these cells into melanocytes (the cells in the skin that can produce the melanin that gives it colour), the researchers were able to find the cause of generalised hyperpigmentation and formation of “café au lait” spots. The loss of neurofibromin expression in these NF1 melanocytes leads to a cascade of dysregulation. First, dysregulation of the AMPc and ERK signalling pathways, followed by dysregulation of the transcription factor MITF, and ultimately, increased expression of the enzymes involved in melanin production.
To correct this, the researchers used pharmacological inhibitors specific for these different signalling pathways, and were able to restore the cells to normal pigmentation levels.
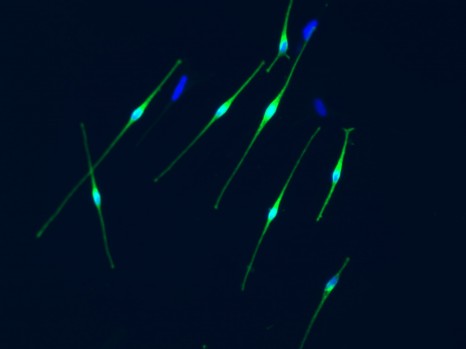
Melanocytes (cells producting melanin) © Inserm/Baldeschi, Christine
Christine Baldeschi
Unité Inserm 861 “Institut des cellules souches pour le traitement et l’étude des maladies monogéniques (I-STEM)”
+ 33 (0) 1 69 90 85 33
rf.metsi@ihcsedlabc
In vitro modeling of hyperpigmentation associated to neurofibromatosis type 1 using melanocytes derived from human embryonic stem cells
Jennifer Allouchea,b, Nathalia Bellonc,d,e, Manoubia Saidanid, Laure Stanchina-Chatroussed, Yolande Massond, Anand Patwardhanf,g, Floriane Gilles-Marsensf,g, Cédric Delevoyef,g,h, Sophie Dominguesd, Xavier Nissand, Cécile Martinata,b, Gilles Lemaitrea,b, Marc Peschanskia,b, and Christine Baldeschia,b
a INSERM U-861, Institut des cellules Souches pour le Traitement et l’Etude des Maladies monogéniques (I-Stem) I-Stem, Association Française contre les Myopathies (AFM), 91030 Evry Cedex, France;
b Université d’Evry Val d’Essonne (UEVE) U-861, I-Stem, AFM, 91030 Evry Cedex, France;
c Department of Dermatology, Reference Center for Dermatologic Diseases, Paris Descartes–Sorbonne Paris Cité University, 75015 Paris, France;
d Centre d’Etude des Cellules Souches (CECS), I-Stem, AFM, 91030 Evry Cedex, France;
e Institut Imagine, Necker-Enfants Malades Hospital, 75015 Paris, France;
f Institut Curie, Paris Sciences et Lettres Research University, F-75248 Paris, France;
g Structure and Membrane Compartments, CNRS UMR 144, F-75248 Paris, France;
h Cell and Tissue Imaging Facility, CNRS UMR 144, F-75248 Paris, France
PNAS, 6 juillet 2015