Leptin is a hormone secreted by adipose cells, sending the satiety signal to the brain. Sometimes, certain overweight or obese individuals develop resistance to this hormone, which is no longer able to deliver its message. A team led by Vincent Prévot, Inserm Research Director at the Jean Pierre Aubert Research Centre in Lille, has used obese mice to show how the pathway transporting leptin to the brain can be reactivated.
Obesity acquired by a diet containing too much fat is associated with high and abnormal levels of circulating leptin. These circulating concentrations reflect the inability of leptin to reach the brain. So it remains ‘trapped’ in the bloodstream. In this study, published in Cell Metabolism, the researchers succeeded in finding the front door for this hormone to enter the brain and restored opening of this ‘cellular door’ in obese mice.
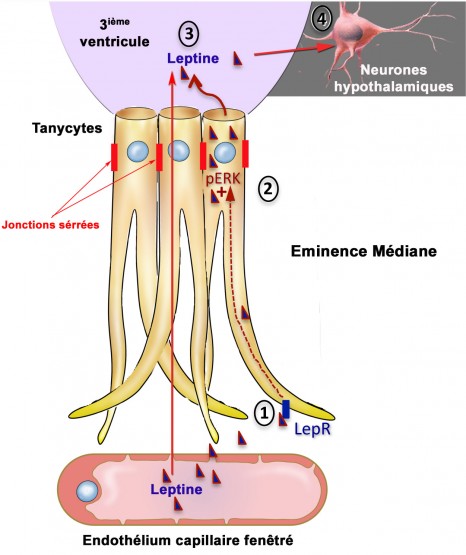
Leptin transport via the tanycytes © Inserm / Vincent Prévot
The research team was first able to describe the mechanism by which leptin passes through this cellular door: tanycytes (see diagram opposite: cells shown in yellow). These cells capture circulating leptin from blood vessels that, at this site, are specially-adapted to allow its passage (step 1). As it passes though the tanycyte, captured leptin activates an ‘ERK’ signalling pathway (step 2) that triggers its release into the cerebrospinal fluid (step 3). Leptin then activates brain regions that convey its anorexigenic action (step 4).
In cases of obesity. the researchers observed that the tanycyte is able to capture the hormone but can no longer release it into the brain. It develops what they call ‘leptin-resistance’ associated with the change in the activity of its receptor, ‘LepR’. The researchers then tested mice with a pharmacological treatment enabling leptin to reach the CSF.
“Our study shows that transport of leptin through the tanycyte can be restored in obese mice using a pharmacological treatment by activating the ERK signalling pathway in this cell“, explains Vincent Prévot, Inserm Research Director.
This treatment allows leptin to activate neurones and finally restore the sensitivity of mice to this hormone by gradually restoring a balanced diet.
“
Altered leptin transport by tanycytes could therefore constitute the first physiopathological event leading to central hormone resistance in overweight or obese individuals” he concludes.
These contents could be interesting :
