Researcher Contact
François PattouDirecteur de l'Unité 1190 « Recherche translationnelle sur le diabète » (Université de Lille, Inserm, CHRU Lille)
03 20 62 69 63
rf.2ellil-vinu@uottapf
Individuals with type 2 diabetes, who are resistant to insulin, have an excess blood glucose level, which they are now trying to reduce using a new class of diabetes drugs known as the gliflozins. These new drugs lower the sugar level but also produce a paradoxical effect, leading to the secretion of glucagon, a supplementary source of glucose. Joint research units 1190, “Translational Research for Diabetes,” (University of Lille, Inserm and Lille Regional University Hospital), directed by François Pattou, and 1011 “Nuclear Receptors, Cardiovascular Diseases and Diabetes,” directed by Bart Staels[1], describe a new mechanism that controls glucagon secretion in humans, making it possible to elucidate this phenomenon and suggesting a modification of this new type of treatment.
These results, obtained in Lille at the EGID (European Genomic Institute for Diabetes) Laboratory of Excellence, are published in the journal Nature Medicine on 20 April 2015.
The team directed by François Pattou is developing innovative therapies to control the more severe forms of diabetes, a disorder characterised by a high blood sugar levels, i.e. chronic hyperglycaemia. To treat type 1 diabetes, the laboratory is conducting projects based on the production of human islets, which are transplanted into patients. Islet transplantation restores production of insulin, the hormone that controls the level of sugar by storing it when its level in the blood is too high. Analysis of human islets destined for transplantation makes it possible to evaluate the cells and thus improve transplantation. It was in this context that the research team discovered a new mechanism for controlling glucagon secretion in humans, a mechanism that explains a side-effect of a new class of diabetes drugs used to treat type 2 diabetes associated with obesity and characterised by insulin resistance.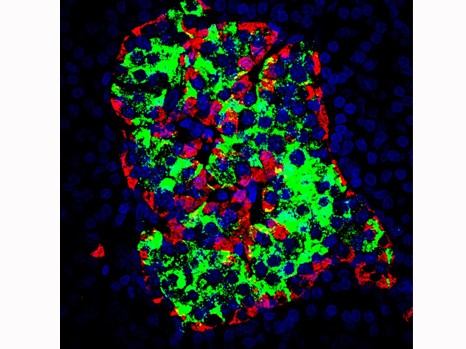
Human islet of Langerhans (0.3 mm diameter) with alpha cells stained red and beta cells stained green
© Inserm Valery GmyrAccessible on www.serimedis.inserm.fr as soon as the embargo is lifted
When the cells detect a low sugar level (e.g. during fasting), an increase in blood sugar level is required to provide the energy needed by the body. This involves another hormone, glucagon, the role of which is to stimulate sugar production by the liver in order to restore the blood glucose levels to normal as quickly as possible. This hormone, secreted by the alpha cells in the islets of Langerhans in the pancreas, has been somewhat forgotten compared to insulin, which is produced by the beta cells to stimulate storage of sugar. It is, however, an essential part of the physiopathology of diabetes.
In this study, the researchers discovered that a glucose cotransporter, SGLT2, known to reabsorb glucose in the kidney, is present in the alpha cells, and controls glucagon secretion. By measuring the expression of the gene for this transporter in the islets of diabetic donors (type 2), they observed a reduction in SGLT2 expression and an increase in glucagon expression compared with the islets of healthy subjects. This result was confirmed in mice with type 2 diabetes. As they became increasingly obese, expression of the cotransporter declined.
Unexpectedly, by revealing this mechanism, the researchers have explained the paradoxical increase in glucagon level observed in patients using a new class of diabetes drugs, the gliflozins, marketed in the United States and the United Kingdom. This class of drugs targets the glucose transporter located in the kidney, preventing the reabsorption of excess glucose in diabetics and its partial elimination in the urine.