Researcher Contact
Patrick Couvreur
Université Paris Sud, Département de la Recherche
01 46 83 53 96
Press Contact
Service de presse de l'Université Paris-Sud
Cécile Pérol
01 69 15 41 99
Service de presse de l'Inserm
©Adobe Stock
A painkiller nanomedicine has just been developed by Patrick Couvreur’s team at Institut Galien Paris-Sud (Université Paris-Sud/CNRS)[i]. This new drug specifically targets the area of painful inflammation without causing the addiction phenomena of current medication.
In pain management, morphine and synthetic opioids are currently effective drugs. Unfortunately, their side effects are serious, in particular respiratory depression, but especially addiction. Opioid addiction is indeed a global healthcare scourge. The United States is particularly affected with 11 million dependent patients and around 175 daily deaths as a result of overdoses.
The use of certain neuropeptides, natural peptides used by neurons to communicate with each other, is undoubtedly an interesting alternative to these drugs. In fact, acting mainly on the neurotransmitter receptors that specifically modulate the pain response function, these natural molecules do not cause these side effects. Unfortunately, after administration these molecules degrade in a few minutes without having time to act.
An effective mode of action without side effects
The “Innovative Nanomedicines for the Treatment of Serious Illnesses” team, led by Patrick Couvreur at the Faculty of Pharmacy at Université Paris-Sud, had the idea of synthesising samples of nanoparticles made up of Leu-enkephalin and coupling them to squalene, a naturally occurring lipid in the body, whose specificity is to be able to provide a protective membrane for the whole compound.
The researchers particularly noted that, unlike morphine, the Leu-enkephalin-squalene nanoparticles spared brain tissue and acted solely on peripheral receptors. Furthermore, imaging showed that the nanoparticles were capable of delivering the neuropeptide specifically to the area of painful inflammation, thus avoiding the central effects responsible for addiction phenomena.
Biochemical and histological investigations conducted on the treated animals, have moreover demonstrated that this new painkiller medicine did not cause any toxicity or side effects.
This study, published in the journal Science Advances, on 13 February 2019, represents a breakthrough in pain treatment due to the nanomedicine. Further research is still necessary before being able to move to clinical trials. A start-up could be established in the coming months to raise the funds needed for their financing.
[i] Together with scientists from the Paris Institute of Psychiatry and Neurosciences (Inserm/Université Paris Descartes) and the Neuropharmacology Laboratory (Université Paris-Sud/Inserm).
Images en cryo-TEM des nanoparticules de Leu-Enképhaline-squalène d’une taille comprise entre 70 et 100 nm.© Institut Galien Paris-Sud, Faculté de Pharmacie
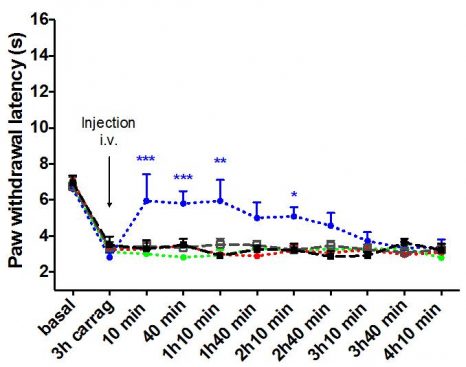
Effet antidouleur des nanoparticules (courbe bleue) aboli après traitement par l’antagoniste naloxone méthiodide (courbe verte) et l’antagoniste naloxone (courbe rouge). Le traitement par la leu-enképhaline libre ou le vecteur seul est sans effet (courbe noire et grise) © Institut Galien Paris-Sud, Faculté de Pharmacie
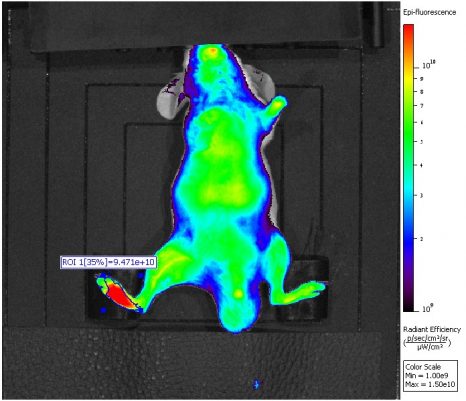
Nanoparticules de Leu-enképhaline-squalène libérant le peptide spécifiquement au niveau du site de la douleur inflammatoire (zone rouge). © Institut Galien Paris-Sud, Faculté de Pharmacie
Patrick Couvreur
Université Paris Sud, Département de la Recherche
01 46 83 53 96
Service de presse de l'Université Paris-Sud
Cécile Pérol
01 69 15 41 99
Service de presse de l'Inserm
Références :
A new painkiller nanomedicine to bypass the blood-brain barrier and the use of morphine,Jiao Feng, Sinda Lepetre-Mouelhi, Anne Gautier, Simona Mura, Catherine Cailleau, François Coudore, Michel Hamon, Patrick Couvreur
Science Advances, février 2019