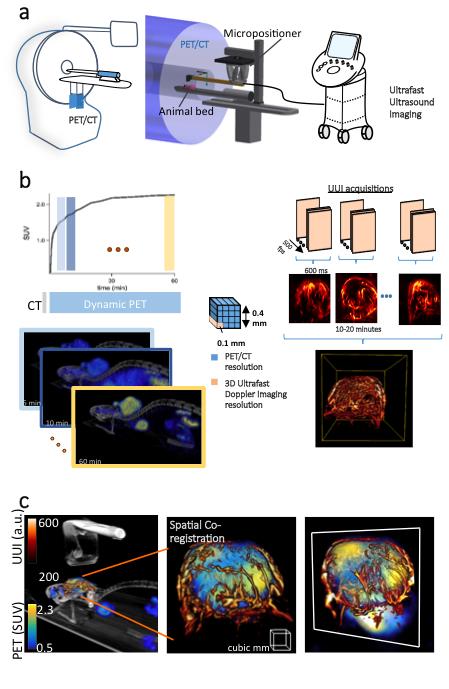
The rapidly-developing medical imaging field could well have found a novel technique in which multiple facets of a living being can be observed in real time and non-invasively. Teams from the Langevin Institute (ESPCI Paris – PSL University / CNRS), the biomedical ultrasound Technology Research Accelerator (Inserm A.R.T.) and the Paris-Cardiovascular Research Center (Inserm / Paris Descartes University) have developed a new medical imaging instrument which combines positron emission tomography – Pet-scan* – with ultrafast ultrasound imaging. Named PETRUS, the acronym of Positron Emission Tomography Registered Ultrafast Sonography, it has obtained 3D images in which organ anatomy, metabolism, function and even elasticity are perfectly superimposed. This research made the cover of the February 6 issue of Nature Biomedical Engineering.
The researchers were able to test their method using commercially-available instruments which were assembled without any major modifications. They imaged cancerous tumors in mice and cardiac activity in rats to test the synchronization of the two methods and the complementarity of the parameters observed. With its ability to visualize several fundamental biological parameters simultaneously in the form of quantitative parametric maps, this new imaging reflects the complex topology of living beings with even greater finesse.
This totally non-invasive technique for observing living beings in real time offers many possibilities, such as to explore the link between the metabolism and the vasculature of organs like the heart, kidney or liver, or to track with greater precision the effect of new cancer treatments, characterize the aftermath of an infarction, etc. Developed in a preclinical setting, PETRUS represents a high-performance and clinically translatable technology for biomedical research.
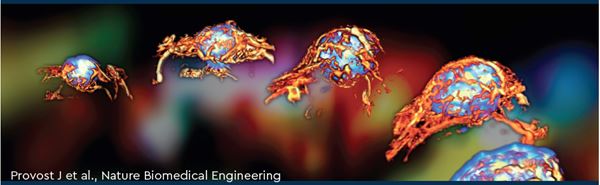
Figure 1: a: PETRUS combines positron emission tomography (PET), computed tomography and ultrafast ultrasound imaging. The three image volumes are correlated using a motorized micropositioner.b: on the left, glucose metabolism kinetics obtained by PET following fluorodeoxyglucose administration; on the right, Doppler 3D ultrafast imaging (500 images per second) showing tumor vasculature.c: imaging of a mouse showing the topography of the metabolic and vascular signals superimposed; on the left, oblique projection; at the center and on the right, enlarged 3D view and slice of a tumor. The color scales shown on the left are the same for the three images. (Provost J et al., Nature Biomedical Engineering)
Figure 2: Closed-thorax PETRUS imaging of a beating rat heart in a short-axis view showing the cardiac anatomy by ultrafast ultrasound (in black and white) and the metabolic activity of the myocardium at end-diastole (left), end-systole (middle) and mid-diastole (right). Note the perfect overlay of the metabolic signal with the myocardial wall. Each image corresponds to the mean signal during one-tenth of a heart cycle. Scale: 1 mm. (Provost J et al., Nature Biomedical Engineering)
*Positron Emission Tomography (Pet-Scan): 3D medical imaging used to visualize the metabolic or molecular activity of an organ, using the principle of scintigraphy.