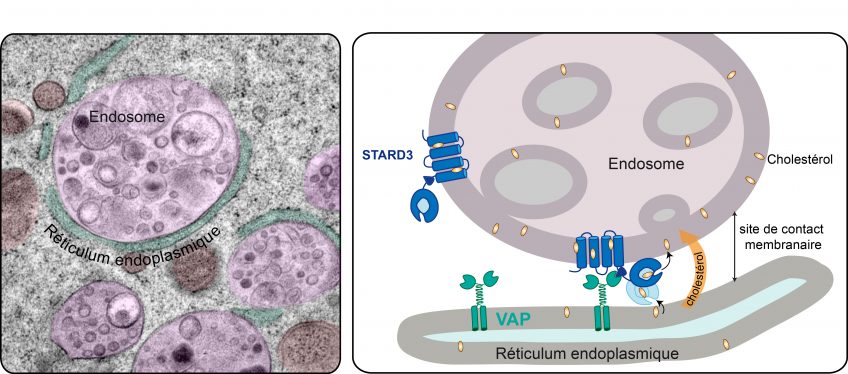
©F Alpy/IGBMC
Cholesterol plays a central role in many living processes. In a new study, a team led by Catherine-Laure Tomasetto, Inserm research director at the Institute of Genetics and Molecular and Cellular Biology (Inserm/CNRS/Université de Strasbourg) reveals the role played by the STARD3 protein in the distribution of cholesterol within cells. A little like molecular velcro, this protein has the capacity to form membrane contacts between two cell organelles, enabling it to transport cholesterol from one organelle to another.
This research has been published in the EMBO Journal
Cholesterol is a component of biological membranes and essential for human cell functioning. A cell has two ways of obtaining cholesterol: by capturing it in the blood and internalizing it using endosomes, or by producing it in the endoplasmic reticulum, a network covering the inside of the cell that synthesizes most lipids. Once captured or synthesized, cholesterol is redistributed throughout the cell’s membranes via mechanisms that have not all been elucidated.
Since cholesterol is not water-soluble, its movements within the cell are very limited. To ensure its transport, the cells have specialized transporters. Catherine-Laure Tomasetto’s team is interested in one of them, protein STARD3, the role of which had remained quite a mystery until now. In this new study, the researchers unraveled some of that mystery. STARD3 is anchored to the endosomes, cell organelles that ensure communication between the outside and the inside of cells. Within the cell, STARD3 attaches to VAP, a protein that is itself fixed to the endoplasmic reticulum. This association creates close appositions between the endosome and the endoplasmic reticulum that are known as membrane contact sites. At these sites, the membranes of the two organelles are very close (less than 30 nm), thus facilitating communication and exchange. In this study, the researchers demonstrated that these membrane contact sites between the endosomes and the endoplasmic reticulum form a type of bridge, enabling STARD3 to transfer cholesterol from the membrane of the endoplasmic reticulum to that of the endosome, thereby rerouting some of the cholesterol that was intended for the plasma membrane.
These results therefore identify a new pathway that regulates cholesterol flow within cells. Understanding how cells balance the two available cholesterol sources will probably help us better understand the mechanisms of certain neurodegenerative or cardiovascular disorders presenting alterations in cholesterol distribution.
This study was funded by the French National Cancer Institute (INCA), the French foundation for medical research (FRM), the French League against cancer, the Ara Parseghian Medical Research Foundation, and the Vaincre les Maladies Lysosomales (overcoming lysosomal diseases) association.
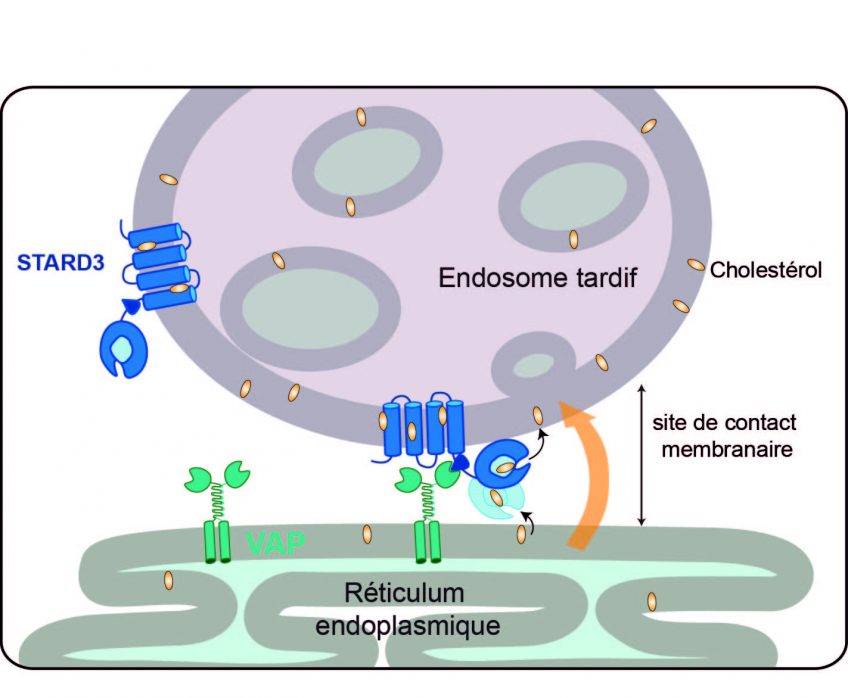
The formation of a membrane contact site for the transport of cholesterol
Image credit: F Alpy/IGBMC