3D illustration of HIV virus © AdobeStock
3D illustration of HIV virus © AdobeStock
Tackling HIV continues to be a major public health challenge, mainly because the persistence of viral reservoirs means that people living with HIV need to take lifelong antiretroviral treatment. But some individuals, known as “post-treatment controllers,” are able to maintain an undetectable viral load even after stopping treatment. In a study funded by ANRS Emerging Infectious Diseases (ANRS MIE), scientists from the Institut Pasteur, Inserm and the Paris Public Hospital Network (AP-HP) identified specific immunogenetic characteristics in a group of these individuals. The research provides novel information about the immune mechanisms associated with HIV control in the absence of antiretroviral treatment and offers new prospects for the development of immunotherapies aimed at achieving remission or a cure for HIV infection. The research was published in Med on April 28, 2025.
Cells infected with HIV persist in the body, forming what are known as viral reservoirs, even in persons receiving antiretroviral therapy. These reservoirs lead to rapid viral rebound if treatment is stopped. But some individuals are able to achieve long-term control of the virus after discontinuing treatment. These “post-treatment controllers” were described in the VISCONTI study in 2013. They are considered to be in long-term virological remission from HIV infection. In some cases, they have been controlling the virus for more than 25 years without treatment. Starting treatment early, within a few days of infection, during the acute infection period, seems to facilitate post-treatment control of HIV,1 but the immune mechanisms had previously remained unclear.
This study, coordinated by Asier Sáez-Cirión, Head of the Institut Pasteur’s Viral Reservoirs and Immune Control Unit, has found that some genetic characteristics associated with innate immunity cells (natural killer or NK cells) are very frequently found in post-treatment controllers in the VISCONTI cohort. A retrospective analysis of the ANRS CO6 PRIMO cohort (where the scientists analyzed the genetic characteristics of more than 1,600 participants monitored since the first weeks following infection) confirmed that the presence of these genetic markers seems to encourage lasting HIV remission in individuals who began treatment early and then stopped it for various reasons. The scientists showed that these genetic markers are accompanied by the existence of specific NK cell populations that are better able to control infection.
“These results support the role of NK cells in long-term HIV remission and could guide the development of novel immunotherapies,” commented Asier Sáez-Cirión.
Ongoing clinical trial
To confirm these findings, a clinical trial entitled ANRS 175 RHIVIERA01, sponsored by Inserm/ANRS MIE, was launched in March 2023. The aim of the trial is to analyze links between the genetic markers of NK cells and post-treatment control. As part of the trial, 16 individuals with these genetic characteristics, who began treatment immediately after infection, were invited to discontinue treatment under close surveillance. The analysis is ongoing. At the same time, the scientists are characterizing exactly how these genetic characteristics associated with remission influence the program and function of NK cells. This approach could pave the way for immunotherapies aimed at mobilizing these specific cells in other people living with HIV.
“This discovery represents a crucial step in efforts to achieve sustained remission from HIV infection. At a time when programs offering access to antiretroviral therapy are coming under threat, there is an urgent need for novel therapies enabling people living with HIV to lead a normal life without the need for treatment,” concludes Asier Sáez-Cirión.
[1] Passaes, et al. Nat Com 2024: HIV: early treatment, one key to remission
This study is associated with the VISCONTI study and the PRIMO cohort, supported and funded by ANRS MIE. The scientists also received funding from the NIH, especially in connection with the ERASE HIV project, which is seeking a cure for HIV.

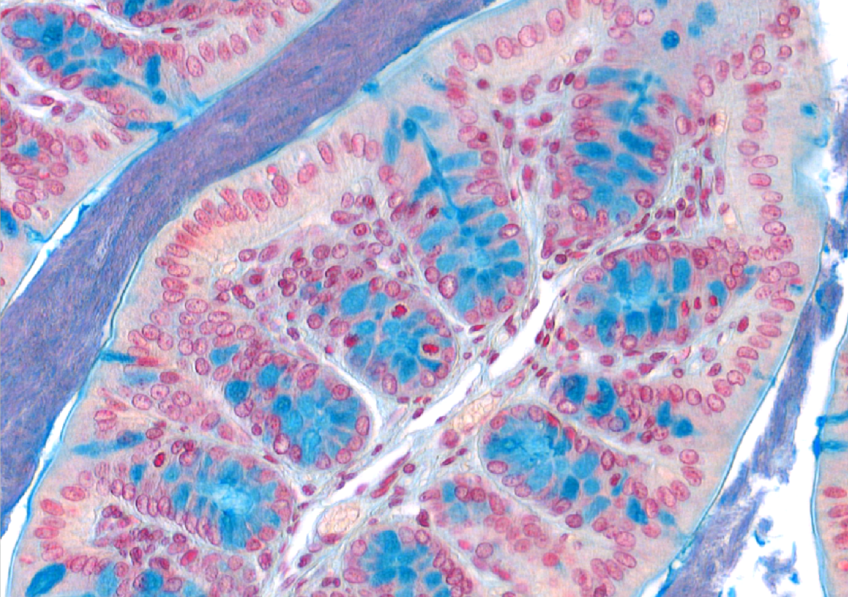
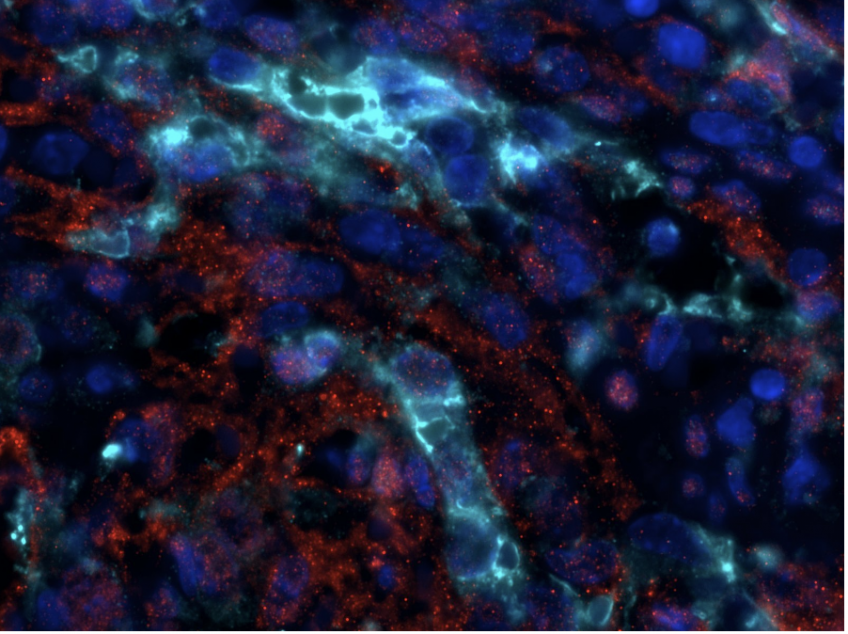 Phospho-ERK (red), Green Fluorescent Protein (cyan) and DAPI coimmunofluorescence on spleen sections from mice carrying a KRAS G12C endothelial mutation © Guillaume Canaud
Phospho-ERK (red), Green Fluorescent Protein (cyan) and DAPI coimmunofluorescence on spleen sections from mice carrying a KRAS G12C endothelial mutation © Guillaume Canaud