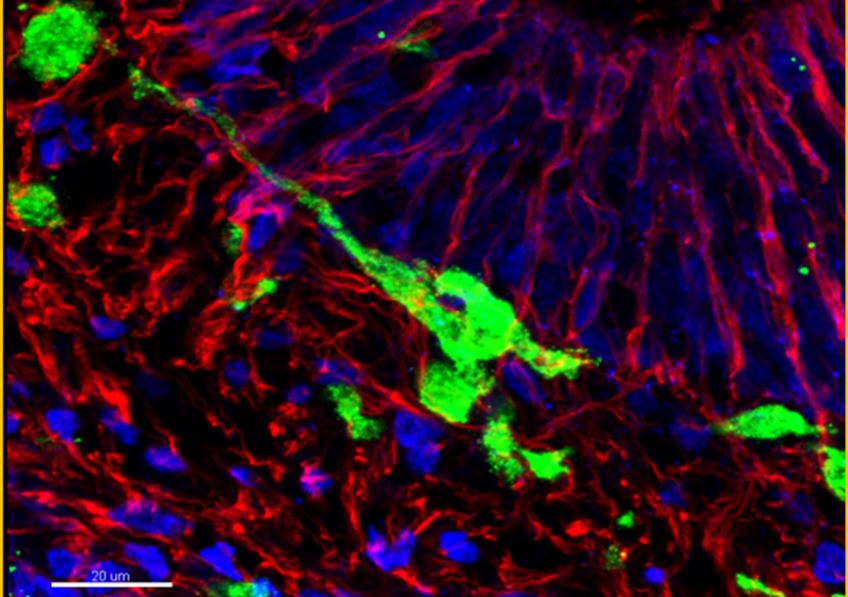
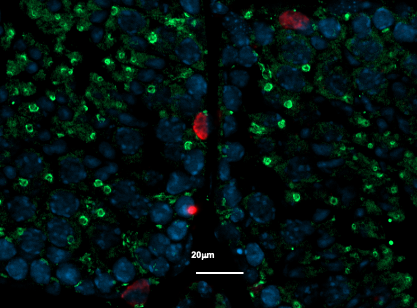
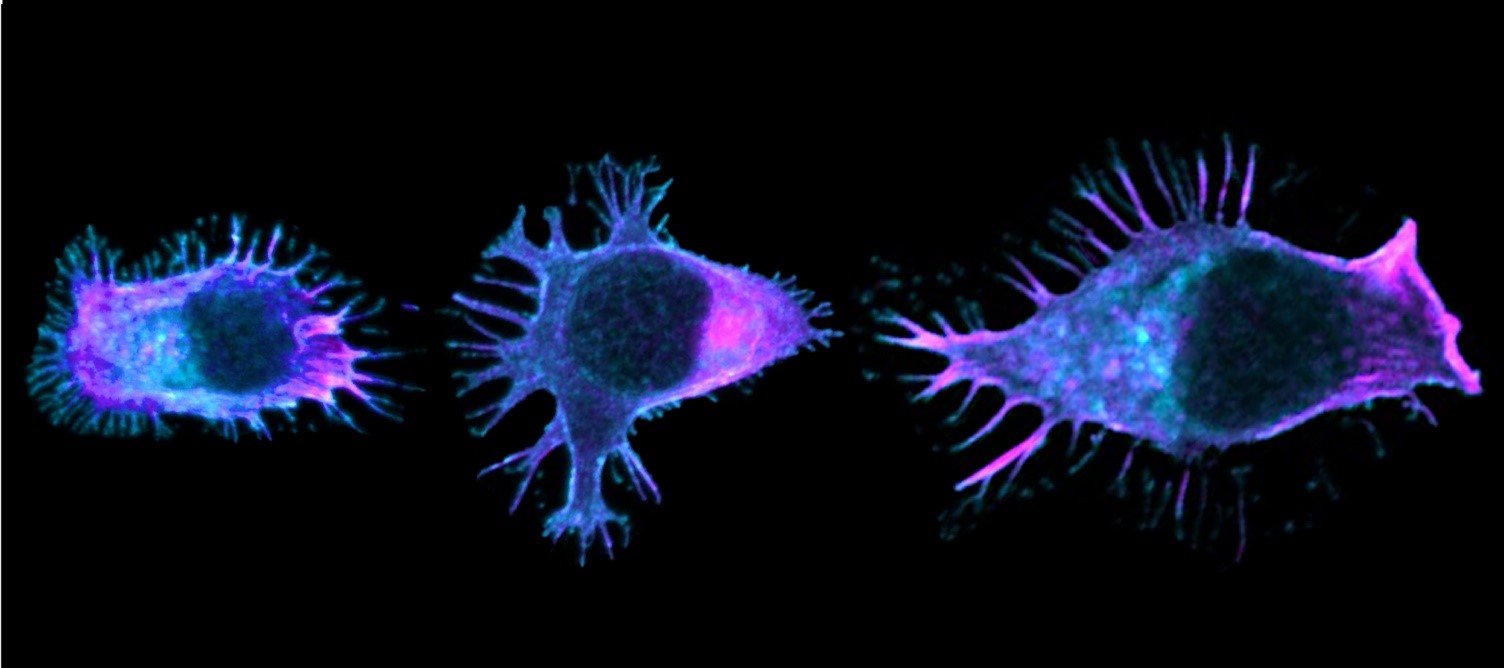
Scanning electron microscopy. Here we see how platelets (in blue/purple) attach to two tumour cells (in red) in a pre-clinical mouse model. © Maria Jesus Garcia Leon (unit 1109 Inserm/Université de Strasbourg)
Scanning electron microscopy. Here we see how platelets (in blue/purple) attach to two tumour cells (in red) in a pre-clinical mouse model. © Maria Jesus Garcia Leon (unit 1109 Inserm/Université de Strasbourg)
What if our blood platelets[1], which play a major role in maintaining the integrity of our circulatory system, were not always on our side? Research teams from Inserm, Université de Strasbourg and the French Blood Establishment have studied their role in the process of metastasis formation. Their findings suggest that platelets, by binding specifically to circulating cancer cells, promote their survival not just in the bloodstream, but also within metastases. This research, published in Nature Communications, also shows that using treatments to target this binding could fight the formation of metastases without the same bleeding risk[2] as with conventional antiplatelets.
A metastasis is a ‘secondary’ tumour which is usually formed from cancer cells that have broken off from a ‘primary’ tumour before migrating through the blood or lymphatic vessels to settle elsewhere in the body. During their migration, these cancer cells encounter the blood platelets – which prove to be unexpected allies. By binding to the cancer cells, the platelets help them to survive the immune cells in the blood environment and exit the bloodstream to reach their metastasis site.
However, not all cancer cells receive the same protection because some bind to the platelets more easily than others. This dictates their capacity to survive in the blood circulation, target certain vascular regions and, as such, their ability to metastasise. Furthermore, in-depth analyses of lung metastases have shown the presence of large numbers of platelets which may play a role that differs from or complements the role they play in blood vessels.
Two teams led by Inserm researchers Jacky Goetz, from the Molecular Immuno-Rheumatology Unit (Inserm/Université de Strasbourg) and Pierre Mangin, from the Biology and Pharmacology of Blood Platelets: Haemostasis, Thrombosis, Transfusion Unit (Inserm/French Blood Establishment/Université de Strasbourg), studied the moments at which the platelets intervened during the migration of cancer cells to promote their survival and spread. They also looked at how to counter this alliance without using conventional antiplatelet drugs which, by altering bleeding cessation, present the risk of haemorrhage.
In a mouse model, the researchers artificially induced controlled falls in the number of platelets at different stages in the formation of lung metastases. They saw that by removing platelets early (while they were still circulating in the blood), they limited the exit from the blood vessel of the cancer cells with a high affinity for the platelets, and thereby inhibited the formation of metastases. Those cancer cells with low levels of platelet binding were also affected, but only when the platelet level was decreased later, when the lung metastases were already formed.
‘These observations suggest that as well as protecting the cancer cells in the bloodstream, the platelets could also protect them against the immune system later on – i.e. within the metastases themselves, helping them to proliferate there, explains Goetz. So the aim of our future research will be to understand how the platelets colonise growing metastases.’
But how can we circumvent the issue of bleeding risk associated with antiplatelet treatments?
One initial avenue could involve a specific protein found on the platelet surface: glycoprotein VI (GPVI). Previous research has suggested that it could modulate the pro-metastatic activity of platelets without causing bleeding. The expression of this protein could be inhibited by glenzocimab, a molecule currently being evaluated in patients for the treatment of stroke. When they used glenzocimab in their animal model, the scientists saw that it effectively reduced the development of already established lung metastases, without affecting the cessation of bleeding.
‘These observations reinforce the idea of the contribution of platelets to the formation of metastases after cancer cells leave the circulation, explains Mangin. In addition, our research highlights the possibilities of developing new therapeutic strategies which, unlike conventional antiplatelet treatments, would not disrupt bleeding cessation and could therefore be considered in oncology, particularly to reduce the progression of lung metastases. Our two teams are currently working to explore this potential,’ adds the researcher.
This study, carried out on experimental animal models, reconciles previous contradictory data on the role of platelets in the metastatic process.
‘Studies in humans, for example on cohorts of patients with long-term exposure to antiplatelet agents for cardiovascular indications, or to evaluate the efficacy of oncology treatments in patients on antiplatelet therapy, could be excellent indicators for verifying these findings,’ concludes Goetz.
[1]Platelets (thrombocytes) present in the blood are not cells per se, but fragments of large bone marrow cells: megakaryocytes. They play a major role in the rapid cessation of bleeding (haemostasis). As such, platelet counts that are too low can lead to clotting disorders and therefore a risk of bleeding (in the event of injury, for example).
[2] See footnote 1