©2019 Flore Avram/Inserm
Today, around 1 in 8 couples seek help because they are struggling to conceive. This is probably linked to the fact that couples are starting families later in life than before, or because they are setting aside the taboos linked to infertility and are more willing to seek help. Infertility has therefore become a public health problem, and the scientific community is rallying in response.
Where are we with research into this area, which lies at the heart of current societal problems? What are the prospects for the transfer of such research into clinical practice? Fertility research covers many different areas. The aim of this press kit is not to tackle them exhaustively, but to highlight the sectors in which research is making progress.
When research makes progress, everyone’s health benefits.
- Research into combating infertility
The term infertility is used when a couple are unable to conceive a child naturally after 12 months of trying. This term covers cases of total sterility, where there is no hope of natural conception, and subfertility, the majority of cases, in which couples have a reduced – but not zero – chance of achieving a pregnancy.
Cases of infertility are divided into 4 categories based on their cause:
– 30% are female-related;
– 30% are male-related. In men, azoospermia and oligospermia are the two leading causes of infertility identified to date;
– 30% are combined, meaning that they are caused by reduced fertility in both partners;
– 10% are unexplained.
In women, with the exception of mechanical causes affecting the fallopian tubes – when they are impaired or blocked (usually following an infection) – or uterus, endometriosis and abnormal ovulation are the most common causes of infertility.
Causes of abnormal ovulation include polycystic ovary syndrome (which affects around 10% of women around the world), hyperprolactinemia, and primary ovarian insufficiency (which may also be a side effect of chemotherapy).
Current research seeks to both improve understanding of the causes of infertility, and also to study new therapies or management methods that aim to increase the chances of conception.
- Improving understanding of the causes
Many researchers are studying the genetic causes of ovarian insufficiency. Several fertility problems are caused by certain genes not working, or not working properly. One rapidly growing area of research, due in particular to the improvement in high-throughput screening methods, is the study of genetic variants.
The Inserm laboratory led by Nadine Binart, for example, is working on primary ovarian insufficiency (POI), which is characterized by the inability of ovarian follicles to mature or by diminished ovarian reserve. Based on DNA analysis of women with POI, researchers are working to isolate the genes that are involved or altered in their genetic make-up. This approach is helping to improve understanding of the disease, but does not make it possible to provide specific treatment to these women, as sterility becomes definitive once there are no more eggs left in their ovaries. Preventive management can however be introduced if the genetic abnormality is found before the ovarian reserve is entirely depleted – for example, during family testing. This is the role of clinical research, which makes it possible to lessen the impact of these diseases when mutations are identified in affected families, to inform young patients about the risk of losing their eggs over time, and to introduce fertility preservation methods if appropriate.
- The hormonal approach: the example of kisspeptin and prolactin
It is well-established that breastfeeding results in increased secretion of prolactin (PRL) by the pituitary gland, inhibiting a woman’s ability to ovulate. This prevents the onset of a new pregnancy. Some diseases also lead to an increase in PRL, including tumors of the pituitary gland from which this hormone is secreted. These cases of hyperprolactinemia, which result in period problems and infertility, are a leading cause of anovulation. In 2011, the Inserm team led by Jacques Young and Nadine Binart revealed the underlying mechanism that blocks ovarian function. Using a mouse model of the disease, the researchers showed that PRL inhibits secretion of a neurohormone called kisspeptin, which is the starting point for the entire hormone cascade responsible for ovarian cyclicity. In a mouse model, administration of kisspeptin made it possible to restore ovarian cyclicity despite the hyperprolactinemia.
This pathophysiological discovery explains the link between infertility and hyperprolactinemia for the first time, and points the way to developing innovative therapies. The basic concept has recently been validated in women,[1] which will make it possible to offer a therapeutic alternative for patients who do not respond to the drugs currently used.
1.2. Preserving fertility: areas of research and latest findings
Specialist “oncofertility” consultations have developed extensively in recent years and should now be an integral part of the care pathway for all young female patients with cancer. Several “fertility preservation” techniques designed to cryopreserve gametes, or preserve reproductive capacity, are now available, and others are currently in development. In France, since 1994, these methods have been included in various pieces of bioethics legislation. Article L.2141 11, modified by law 2011-814 of July 7, 2011, states that “All persons whose fertility is likely to be impaired by their medical care, or whose fertility risks being prematurely impaired, may have their gametes or reproductive tissue collected and preserved with a view to their later use of assisted reproductive technology, or with a view to preserving and restoring their fertility.” Fertility preservation methods are also included in the 2014-2019 Cancer Plan, which stipulates that “all patients must have access to cancer treatments, and innovative treatments in particular.”
- Improving gamete preservation
Several techniques for cryopreserving female gametes are currently available. The standard method involves freezing mature eggs or embryos obtained from these eggs. It is not however suitable for prepubescent girls, who need to begin treatment urgently, and can also present problems in patients with hormone-sensitive cancers. Therefore, other techniques, although still considered experimental, may be offered in these situations.
Improving the available methods and developing new strategies is currently a major focus for oncofertility. This is one of the areas of research on which the Inserm team led by Nadine Binard and Charlotte Sonigo is working, in collaboration with Prof. Michael Grynberg.
- Using anti-Müllerian hormone
Chemotherapy reduces fertility through a direct toxic effect on the ovaries. Cyclophosphamide, which is commonly used in cancer treatment, causes massive destruction of the germ cells contained in the ovarian follicles. In a mouse model, researchers have recently shown that treatment with anti-Müllerian hormone, which is normally secreted by the ovaries, can limit reduction of follicular reserve during chemotherapy. Use of anti-Müllerian hormone is therefore a promising approach to fertility preservation.
1.3. The role of new technologies: Using artificial intelligence in reproductive research
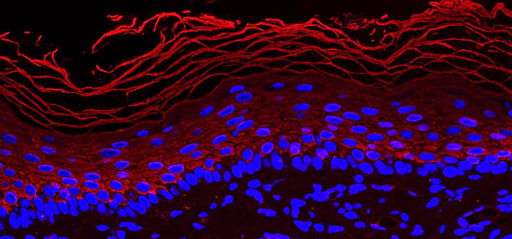
The store of germ cells contained in the follicles constitutes the ovarian reserve. Assessing the quantity of these germ cells is a common way of providing information on ovarian physiology and of measuring the impact of the environment on the ovaries. The standard method used in mice is time-consuming and tedious. In conjunction with a company specializing in artificial intelligence, Inserm researchers have recently developed an automated artificial intelligence method for follicle counting that uses a deep learning approach.[2] This new tool will be made available to the fertility research scientific community, saving a great deal of time and enabling better reproducibility of data.
- Research into combating endometriosis
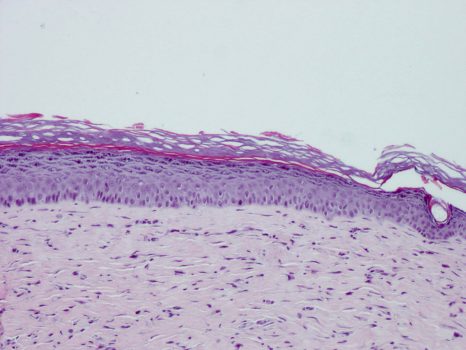
Endometriosis is a complex disorder characterized by chronic inflammation due to the presence of tissue resembling the uterine lining outside the uterus. This “ectopic uterus” continues to respond to ovarian hormones, which in some women can cause severe pain and sometimes infertility. In response to increased visibility of the disease in the media, notably due to the work of patient organizations, the French health minister has announced an action plan to improve management of endometriosis. In terms of research, there has been a surge in studies of endometriosis over the last 5 years. Around 1,200 articles per year are being produced by researchers around the world, helping to advance understanding of this disorder.
©2019 Flore Avram/Inserm
- An estimated 1 in 10 women have some form of endometriosis.
- The locations of endometriosis lesions vary.
- Endometrial cell reflux during periods occurs in 90% of women, but only 10% of them develop disease.
- The disease is typically described as having 4 stages, based on the extent and depth of lesions; however, there is no correlation between disease symptoms and severity.
- There are 3 forms of endometriosis: superficial peritoneal endometriosis, ovarian endometriosis (or endometrial cyst, or endometrioma), and deep endometriosis.
2.1. Improving understanding of the causes
The epidemiological approach
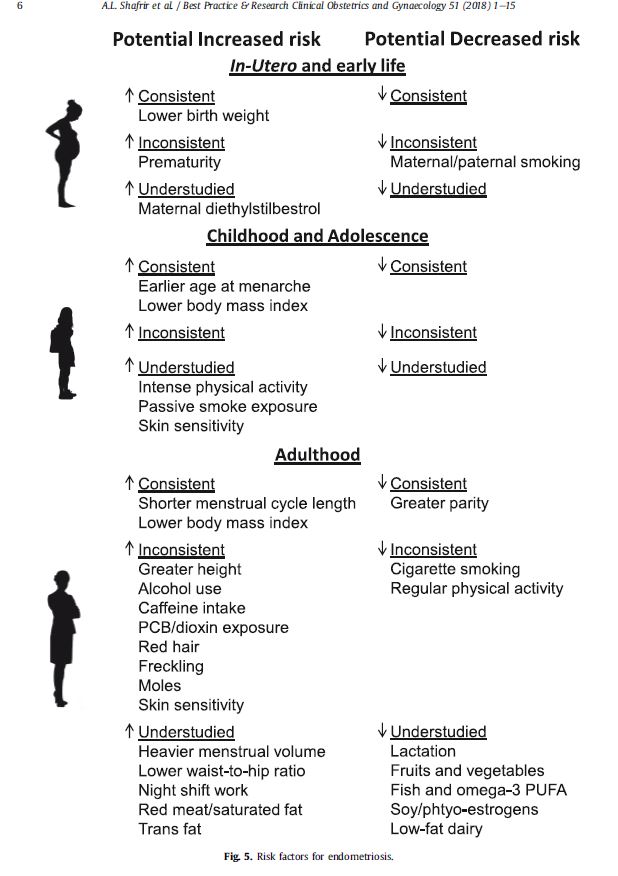
At present, little is known about the causes of endometriosis, its natural history, and the factors affecting its progression. Epidemiological research plays a crucial role in advancing knowledge in this area. There are only a few large epidemiological cohorts around the world in which these aspects can be explored. The largest cohort for exploring endometriosis risk factors is currently a cohort of 116,430 American female nurses who were between 25 and 42 years old in 1989. The risk factors identified in the literature and confirmed in this cohort include: low birth weight, early menstruation, low body mass index, and short menstrual cycles (under 24 days).[3] However, beyond these factors, little information is available on the causes of the disease, and its natural history is largely unknown. The following table is based on a review of the literature published in August 2018:

*The positive association between smoking and reduced risk of endometriosis may be explained by the antiestrogenic effect of tobacco. This would confirm the therapeutic interest of estrogen blockers, which are available in drug form: far more suitable than cigarettes, whose harmful effects have been widely documented.
In a bid to improve understanding of this disease, several epidemiological studies are being launched in France by the team led by Marina Kvaskoff, Inserm epidemiologist and researcher. These include a recently formed patient cohort dedicated to the study of endometriosis: the ComPaRe-Endometriosis cohort. The study team’s objective is to have enough women in the cohort to obtain robust findings in relation to the many questions that are still unanswered about this disease. In less than 6 months, over 8,000 women have already taken part in the study. The team aims to recruit 15,000 to 20,000 participants, and a broad call for participation has gone out to women with endometriosis or adenomyosis (a form of endometriosis limited to the muscle wall of the uterus) to help speed up research into these disorders simply by completing online questionnaires about their experience of the disease (https://compare.aphp.fr/). The study initially looks to explore the natural history of the disease (change in the symptoms and characteristics of the disease over time), and to identify the factors that determine its progression and result in better response to treatment. This research will also make it possible to describe the circumstances of diagnosis and the patient care pathway, and to assess the impact of the disease on patients’ daily lives.
Endometriosis is also being studied within large French cohorts, such as the CONSTANCES cohort, a prospective study of 200,000 men and women (105,000 women) representative of the French population. Marina Kvaskoff’s team has developed an epidemiological research study to determine the prevalence and incidence of the disease in France, and to explore its risk factors within this cohort. Other studies are currently in development and will be conducted in other cohorts in due course.
- The environmental approach
Several epidemiological studies have explored the associations between organochlorine chemicals (solvents, pesticides, insecticides, fungicides, etc.) and endometriosis, but their results have been inconsistent. A French meta-analysis of 17 studies[4] published in February 2019 attempted to draw more robust findings. The risk of developing endometriosis was 1.65 times higher in women exposed to dioxins, 1.70 times higher for those exposed to polychlorinated biphenyls (PCB), and 1.23 times higher for organochlorine pesticides. Although statistically significant, these estimates should be considered with caution due to the significant heterogeneity of the studies and the small estimated effect size. The level of evidence was judged to be “moderate” with a serious risk of bias, supporting the need to conduct further well-designed epidemiological research in order to fill the persistent data gaps.
- Using the genetic and epigenetic approach for early detection
Detecting endometriosis in the early stages, before patients experience symptoms, would make it possible to improve patient care. Although the heritability of endometriosis has been estimated at 50%, it is highly complex and clearly highly polygenic. Numerous candidate genes have been studied from this perspective in analyses of disease predisposition. Initial results have shown that there is no gene for endometriosis, but that the existence of genetic variants characteristic of the disease could enable it to be diagnosed and to improve patient care. In 2017, efforts by the international community made it possible to identify a total of 14 variants (located on the genes WNT4, GREB1, ETAA1, IL1A, KDR, ID4, CDKN2B-AS1, VEZT, FN1, CCDC170, SYNE1, FSHB, and in the chromosomal regions 7p15.2 and 7p12.3). These 14 genes are involved in proliferation and the cell cycle, adhesion and the extracellular matrix, andinflammation, which makes sense in relation to endometriosis. However, each of the variants identified explain only a small part of the genetic variation in endometriosis. In future, the combination of high-risk alleles in a patient might provide a probability of being affected that could be used to diagnose patients and categorize them based on endometriosis type and severity.
The existence of specific epigenetic markers for endometriosis could also theoretically be used for early detection, with endometrial cells presenting specific epigenetic abnormalities that modify expression of the main transcription factors. However, it is not known how the interactions between the defective epigenomic cells and mutated epithelial cell genes contribute to the pathogenesis of endometriosis.
The full complexity of endometriosis cannot however be understood through genetics alone. Genes only influence phenotype through their expression. This expression is regulated by epigenetic molecular mechanisms. As such, most research focuses on studying the microRNA that could be “markers” for the disease. Several have been identified in patients’ plasma thus far, but with very poor reproducibility from one research team to the next. For example, a study published in 2013[5] identified just four miRNA (miR-199a, miR122, miR145*, and miR-542-3p) as enough to categorize patients, with very few errors. Confirmation of this article’s findings in independent cohorts has however been slow. One possible explanation for this is the fact that extraction of circulating RNA remains very heterogeneous from one study to the next, perhaps due to the technical tools used in extraction. In future, new, more comprehensive approaches could provide more consistent results.
- The cellular approach: oxidative stress
Several studies have shown increased oxidative stress in the serum of women with endometriosis. Oxidative stress is a highly general mechanism that induces and is caused by inflammation. It would seem logical to find changes linked to oxidative stress in the context of a painful disease like endometriosis. In mouse models, treatment with antioxidants (N-acetylcysteine) has been seen to reduce endometrial lesions.
Research led by a team from the Institut Cochin has also identified several genes linked to glutathione metabolism within the gene cascades that are deregulated in endometriosis lesions. Glutathione is a peptide that plays a key role in detoxification of hydrogen peroxide, a central molecule in oxidative stress. Down-regulation, particularly of the GCLM and GCLC genes crucial to glutathione synthesis, could explain increased oxidative stress in endometriosis lesions.
- The dysfunctional immune system: a possible approach?
The survival of endometrial cells outside the uterus could be linked to poor function of the immune system causing chronic local inflammation. Studies have shown an increase in some immune cells around endometriosis lesions.
2.2. Treatment: areas of research and latest findings
Changing diagnostic methods: phasing out surgery
Before considering treatment, the first stage is to reduce the diagnosis time for endometriosis, which is currently around 7 to 10 years after onset of the initial symptoms. With this in mind, doctors and researchers are working to develop a diagnostic score, based on a dozen questions, from which doctors will be able to provide a diagnosis with 85-90% reliability. This score may be accompanied if necessary by imaging, which can inform endometriosis diagnosis if carried out and interpreted by trained medical personnel.
Doctors and researchers agree that diagnostic surgery is contraindicated for endometriosis.
The 3 pillars of treatment
Drug therapy, surgery, and assisted reproductive technology (ART) are currently the only 3 methods for treating the symptoms of endometriosis and its potential impact on fertility. In the absence of new treatments, the key is to understand the role played by each component in this therapeutic arsenal, so that they can be used effectively.
Drug therapy is based on blocking ovarian function to bring about artificial menopause via continuous administration of contraceptives. These therapies (the combined pill, estrogen pill, or GnRH agonists) must be personalized and adapted to the needs of the patient. These therapies should be prescribed as the first-line treatment for women who are not seeking to become pregnant, in order to reduce the pain caused by the disease.
For patients who want to conceive, ART and surgery may be considered. ART should be used routinely before all surgical procedures in order to maximize the chance of conceiving a child for couples who want to do so. Surgery must not be used in women who do not want to conceive in whom drug therapy is effective. Endometriosis surgery can be highly invasive and debilitating (removing some parts of the colon, with a high risk of ovarian reserve depletion if ovarian cysts are removed, etc.), and does not prevent the disease from recurring, as it does not treat the cause. Doctors and researchers also agree that women who undergo surgery at a young age have a high risk of their endometriosis lesions recurring, and encounter further difficulties if they decide they want to become pregnant.
All efforts must therefore be made to move away from using surgery as the standard treatment for endometriosis, as has too often been the case in the past.
Some forms of endometriosis – particularly those affecting the ovaries – are now an indication for providing women with access to various fertility preservation techniques.
The role of new technologies: the example of high-intensity focused ultrasound
In Lyon, teams of research clinicians led by Prof. Gil Dubernard (Hospices Civils de Lyon and Inserm unit 1032 LabTAU) have developed an ultrasound-based treatment for bowel endometriosis. When endometriosis infiltrates the rectal wall, it causes debilitating rectal pain that may affect quality of life. After failure of medical treatment, a surgical procedure is often proposed that consists of removing all or part of the rectum and sometimes requires a temporary colostomy (artificial anus).
A phase I clinical trial carried out in 11 patients in 2017 demonstrated that high-intensity focused ultrasound may be a useful alternative to surgery. An ultrasound probe inserted into the rectal passage is able to “desensitize” the lesions within a few minutes. A follow-up trial of 12 patients seeking to confirm these initial results was completed on April 1, 2019. Data analysis is ongoing and will be available shortly.
Meanwhile, in collaboration with the company EDAP TMS (the clinical trial sponsor), the therapeutic ultrasound Inserm laboratory led by Cyril Lafon, LabTAU (Université Claude Bernard Lyon 1/Inserm), is working on optimizing the conditions of ultrasound delivery (insonification) and improving the ergonomics of the probe in order to increase the number of patients eligible for this new treatment.
It is highly likely that this innovative therapy will replace many of the rectal surgeries carried out in this functional disorder that resolves upon menopause.
[1] Hypothalamic-Pituitary-Ovarian Axis Reactivation by Kisspeptin-10 in Hyperprolactinemic Women With Chronic Amenorrhea.
Millar RP, Sonigo C, Anderson RA, George J, Maione L, Brailly-Tabard S, Chanson P, Binart N, Young J.
[2] Sonigo C, Jankowski S, Yoo O, Trassard O, Bousquet N, Grynberg M, Beau I, Binart N. High-throughput ovarian follicle counting by an innovative deep learning approach. Sci Rep. 2018 Sep 10;8(1):13499. doi: 10.1038/s41598-018-31883-8.
[3] https://www.ncbi.nlm.nih.gov/pubmed/30017581
[4] https://www.ncbi.nlm.nih.gov/pubmed/30530163
[5] (Wang et al., JCEM, 2013)