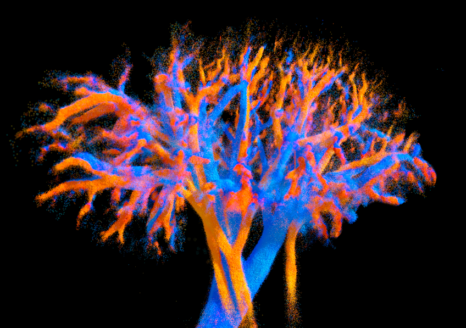
 4D visualisation of the vascularisation of an entire kidney obtained using the multi-lens probe developed in this study. Veins are shown in blue and arteries in red. Colour variations indicate blood flow velocity: the brighter the colour, the faster the blood is flowing. The smallest vessels are less than 100 micrometres in diameter. © Alexandre Dizeux, Physics for Medicine/Inserm
4D visualisation of the vascularisation of an entire kidney obtained using the multi-lens probe developed in this study. Veins are shown in blue and arteries in red. Colour variations indicate blood flow velocity: the brighter the colour, the faster the blood is flowing. The smallest vessels are less than 100 micrometres in diameter. © Alexandre Dizeux, Physics for Medicine/Inserm
For the first time, a team of Inserm researchers from the Institute of Physics for Medicine Paris (Inserm/ESPCI Paris-PSL/CNRS) has succeeded in mapping the blood flow of an entire organ in animals (heart, kidney and liver) with great precision, in four dimensions: 3D + time. This new imaging technique, when applied to humans, could both improve our understanding of the circulatory system (veins, arteries, vessels and lymphatic system) and facilitate the diagnosis of certain blood circulation-related diseases. These results are published in Nature Communications.
Blood microcirculation is a complex network that transports blood to tissues and organs through tiny blood vessels. When this mechanism functions properly, cells receive the oxygen and nutrients they need to stay healthy, while metabolic waste products are efficiently removed.
Any alteration in this network, whether structural or functional, can have serious clinical consequences, including heart failure, kidney failure and various chronic diseases. However, there is currently no imaging method that can visualise microcirculation and assess the integrity of the entire circulatory system, from the large arteries to the finest arterioles, at the level of the whole organ.
With this issue in mind, the Institute of Physics for Medicine (Inserm/ESPCI Paris-PSL/CNRS) research team has developed the first tool capable of producing these images . It is a new type of ultrasound probe, developed as part of Nabil Haidour’s thesis work, under the supervision of Clément Papadacci (Inserm researcher). Using this technology, scientists have been able to map the vascularisation and quantify the blood flow dynamics of three essential organs – the heart, kidney and liver – in animal models of comparable size to humans, all with unprecedented image resolution.
The non-invasive device made it possible to distinguish microcirculation even in the finest vessels (less than 100 micrometres). In the case of the liver, it was possible to identify and differentiate its three blood networks (arterial, venous and portal) thanks to their haemodynamic signature.
‘The originality of these results lies in the fact that these images allow us to visualise the vessels of an entire organ at very small scales (less than 100 micrometres) – this 4D image resolution is unprecedented, as is the ability to observe an entire large organ and its flow dynamics,’ explains Clément Papadacci, Inserm researcher and last author of the study.
This technology will now be tested in humans as part of a clinical trial. Developments enabling deployment in humans are being carried out with the help of the Technological Research Accelerator for Biomedical Ultrasound, created by Inserm and integrated into the Physics for Medicine Institute.
‘The probe can be connected to small portable equipment, which would allow it to be integrated into medical practice,’ explains Clément Papadacci.
‘Used in clinical settings, this new technology could become a major tool for better understanding vascular dynamics as a whole, from the largest vessels to the pre-capillary arterioles. It could also help advance the diagnosis of microcirculation disorders and the monitoring of treatments for small vessel diseases, which are complex to diagnose and are diagnosed by ruling out other pathologies,’ concludes Clément Papadacci.
Illustration of this new technology in video :
[1] Research conducted as part of the MicroFlowLife study (ERC starting grant).



 © AdobeStock
© AdobeStock