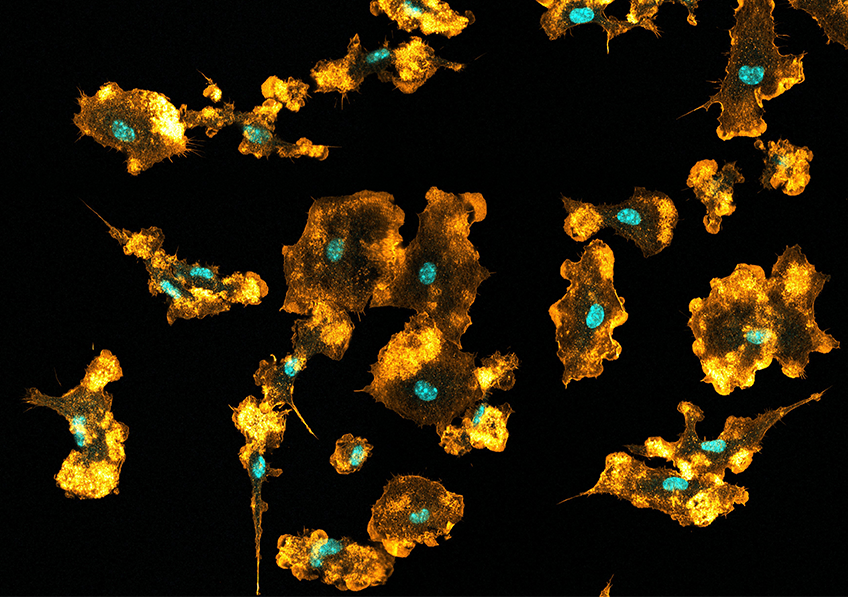
 Image de microscopie à fluorescence de corps cellulaires des nocicepteurs innervant la peau. Les points verts représentent un ARN des nocicepteurs non-peptidergiques activés. © Lilian Basso/Inserm
Image de microscopie à fluorescence de corps cellulaires des nocicepteurs innervant la peau. Les points verts représentent un ARN des nocicepteurs non-peptidergiques activés. © Lilian Basso/Inserm
Les troubles dermatologiques sont caractérisés par une inflammation et des démangeaisons sévères, et les traiter ainsi que les soulager représentent un véritable défi pour la recherche. Les mécanismes par lesquels certains neurones sensoriels, appelés nocicepteurs, intègrent les stimuli en périphérie du corps pour traiter simultanément l’inflammation et la démangeaison sont restés inexplorés pendant de nombreuses années. Dans une étude parue dans le journal Immunity, une équipe de l’Inserm, du CNRS et de l’Université de Toulouse, a montré que dans la dermatite de contact (une inflammation cutanée aiguë à l’origine de démangeaisons importantes), deux catégories de nocicepteurs avaient des fonctions biologiques distinctes. En effet, l’une permettrait de réguler les démangeaisons, tandis que l’autre semble agir directement sur l’inflammation provoquée par certains troubles dermatologiques. Ces résultats ouvrent la voie à de nouvelles stratégies thérapeutiques pour améliorer la vie des patients atteints de troubles dermatologiques.
La dermatite de contact (DC) est une maladie cutanée aiguë courante qui affecte environ 20 % de la population, et se caractérise par une hypersensibilité aux allergènes chimiques, une inflammation, et des démangeaisons sévères. Ces dernières résultent de l’activation dans la peau de terminaisons sensitives de plusieurs populations distinctes de neurones sensoriels regroupées sous le nom de nocicepteurs. Leur rôle est de transmettre la douleur et les démangeaisons au cerveau en passant par la moelle épinière, déclenchant des réponses comportementales comme les grattements.
Récemment, il a également été mis en évidence que les nocicepteurs sont capables de réguler la réponse de nombreux types de cellules immunitaires impliqués dans la dermatite de contact, notamment des neutrophiles (globules blancs qui détruisent les pathogènes). Cependant, le rôle individuel des différentes populations de nocicepteurs dans le développement de la DC restait jusqu’à maintenant inconnu.
Dans une étude dirigée par Nicolas Gaudenzio et Lilian Basso, chercheurs Inserm à l’Institut Toulousain des Maladies Infectieuses et Inflammatoires (Inserm/CNRS/Université de Toulouse), les scientifiques ont pu identifier que différents circuits neuronaux contrôlent indépendamment la réponse inflammatoire et la démangeaison dans la DC.
L’équipe de recherche a combiné des approches génétiques et pharmacologiques dans un modèle de DC chez la souris. En isolant les neurones innervant spécifiquement la peau lors de l’inflammation cutanée, elle a comparé cellule par cellule leurs séquences d’ARN et a pu identifier deux populations de nocicepteurs activés de façon différentes : une population de nocicepteurs dits « non-peptidergiques », qui subissaient un changement au niveau de l’ensemble de leurs ARN, ainsi qu’une autre population de nocicepteurs dits « peptidergiques », qui ne subissait que quelques changements discrets.
Les chercheurs ont pu ensuite observer que la diminution des nocicepteurs peptidergiques au niveau d’une lésion cutanée était associée à une forte augmentation de l’inflammation avec une production locale accrue de neutrophiles, sans apaisement de la sensation de démangeaison. Au contraire, la diminution de la population de nocicepteurs non-peptidergiques était associée à une diminution drastique des démangeaisons sans aggravation de l’inflammation.
De façon surprenante, le rôle anti-inflammatoires des nocicepteurs peptidergiques semble en opposition avec leurs effets habituellement plutôt pro-inflammatoires dans d’autres contextes de pathologies cutanées, comme la dermatite atopique ou le psoriasis. « Les nocicepteurs peptidergiques semblent intervenir dans la régulation de la réponse immunitaire en limitant la production locale de neutrophiles, indique Lilian Basso. Cette capacité semble être fortement liée au contexte inflammatoire dans lequel ils sont activés, mais aussi au rôle des neutrophiles dans ce contexte : s’il est bénéfique, les nocicepteurs auront une action pro-inflammatoire, et s’il est néfaste, leur action sera plutôt anti-inflammatoire ».
L’équipe de recherche a aussi pu constater une fonction cicatrisante de la population de nocicepteurs non-peptidergiques dans les tissus cutanés lésés. « Il est connu que la cicatrisation des plaies, également présente dans les pathologies inflammatoires cutanées, s’accompagne de démangeaisons. Notre étude suggère que cette sensation de démangeaison pourrait être en fait le témoin de la contribution à la cicatrisation tissulaire des nocicepteurs non-peptidergiques » explique Tiphaine Voisin, chercheuse Inserm et première autrice de l’étude.
Ces résultats révèlent donc la présence de deux circuits neuronaux distincts et adaptatifs, qui réguleraient indépendamment l’inflammation via les nocicepteurs peptidergiques, et les démangeaisons de la peau via les nocicepteurs non-peptidergiques.
« Dans l’ensemble, nos résultats soulignent l’importance de considérer la plasticité des nocicepteurs lors de troubles dermatologiques afin de permettre une analyse plus précise des circuits neuronaux mis en jeu lors de la régulation de différentes manifestations de certaines pathologies. Cette nouvelle compréhension nuancée de la façon dont les nocicepteurs contribuent à l’inflammation et à la démangeaison pourrait ouvrir la voie à des stratégies thérapeutiques innovantes pour gérer simultanément l’inflammation et la démangeaison dans la DC et d’autres troubles cutanés », conclut Nicolas Gaudenzio.

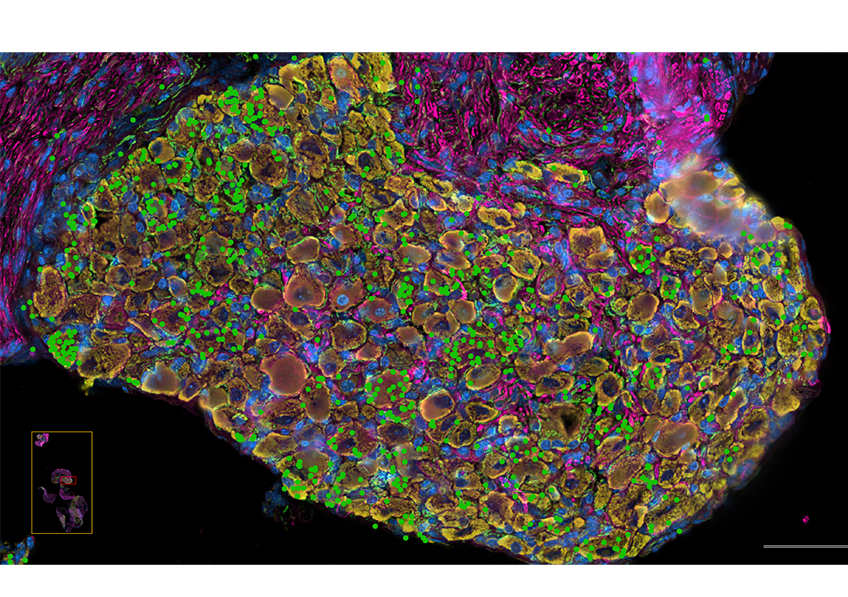
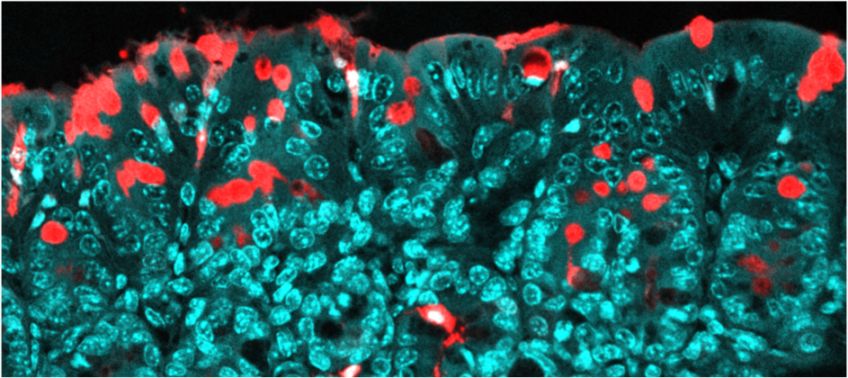 Molecules passing through the colon epithelium via specialized pathways (goblet cells). In red: the molecules transported (antigens); in blue: the epithelium. © Institut Pasteur/Microbiome-Host Interactions/Clara Delaroque and Benoit Chassaing
Molecules passing through the colon epithelium via specialized pathways (goblet cells). In red: the molecules transported (antigens); in blue: the epithelium. © Institut Pasteur/Microbiome-Host Interactions/Clara Delaroque and Benoit Chassaing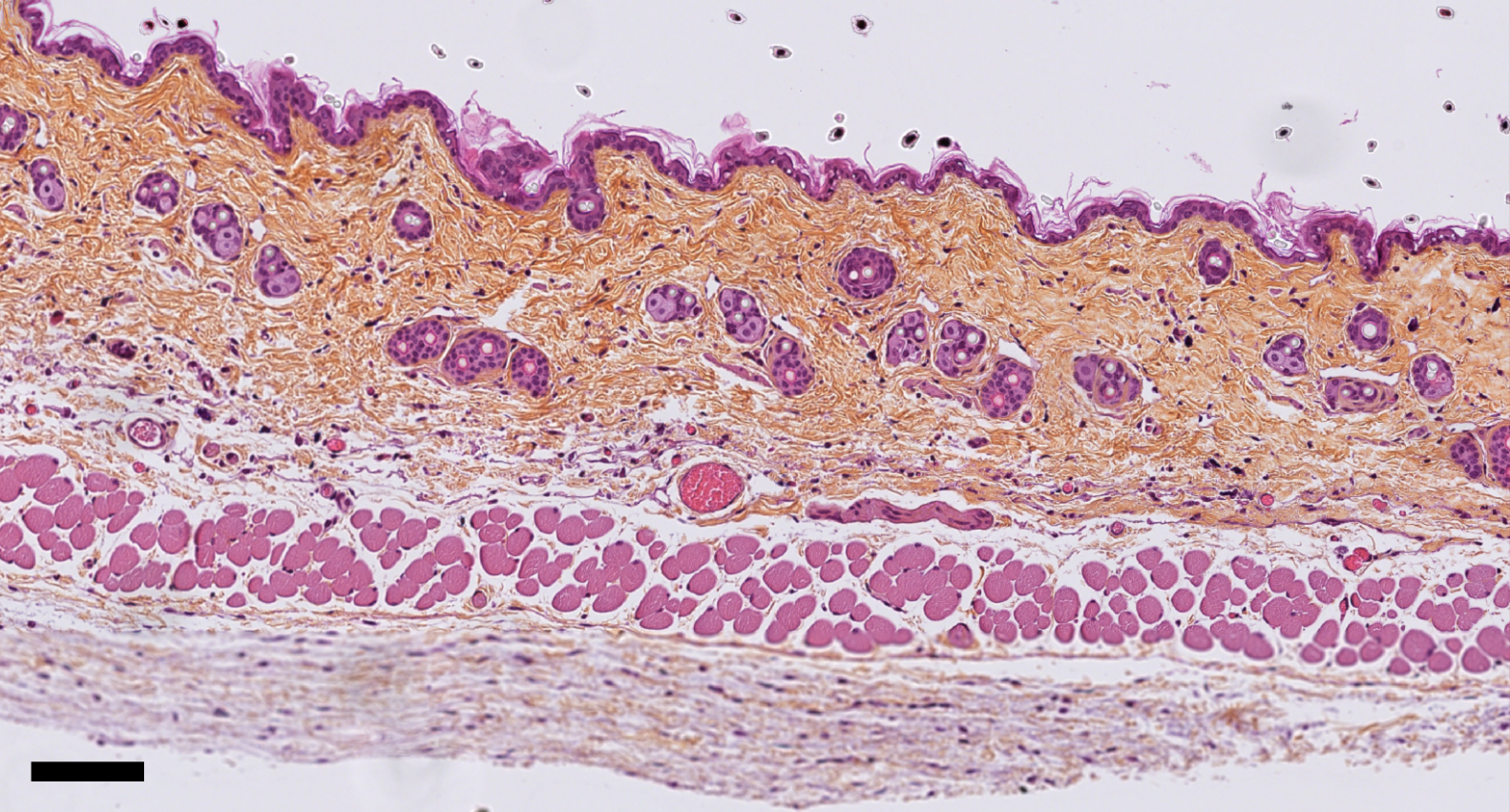
 Mouse skin structure after stretching, using histological staining. The scale bar corresponds to 100 micrometers. © Darawan Tabtim-On and Renaud Leclère – Experimental Pathology Platform, Institut Curie
Mouse skin structure after stretching, using histological staining. The scale bar corresponds to 100 micrometers. © Darawan Tabtim-On and Renaud Leclère – Experimental Pathology Platform, Institut Curie