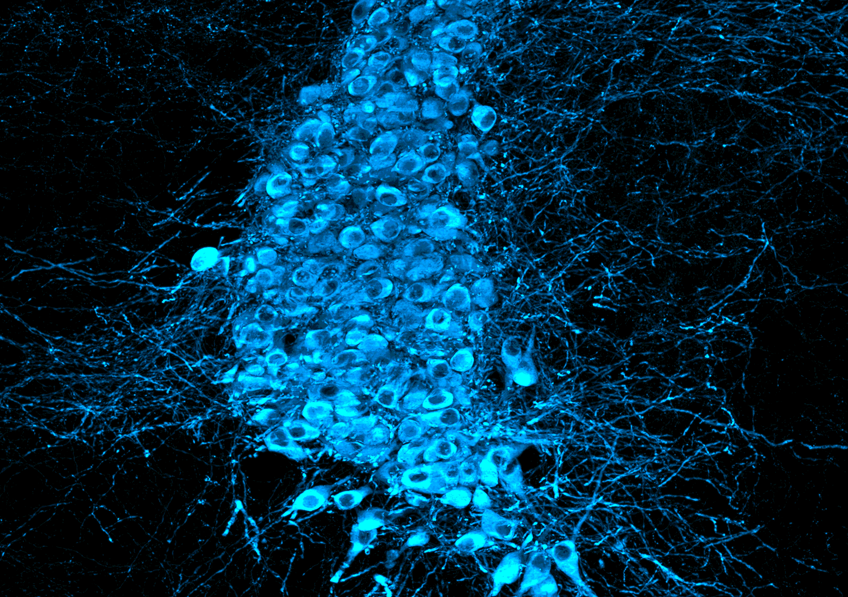
 Tanycytes (white) capture Tau protein (red) circulating in the cerebrospinal fluid and transport it along their extensions/arms, which pass through brain tissue and come into contact with blood vessels (green), into which they release this protein, which is involved in Alzheimer’s disease when it accumulates in the brain. © Vincent Prévot/Inserm
Tanycytes (white) capture Tau protein (red) circulating in the cerebrospinal fluid and transport it along their extensions/arms, which pass through brain tissue and come into contact with blood vessels (green), into which they release this protein, which is involved in Alzheimer’s disease when it accumulates in the brain. © Vincent Prévot/Inserm
Patients with Alzheimer’s disease have common biological alterations, including abnormal accumulation of the Tau protein in the brain. The mechanisms behind this abnormality may be on the verge of being elucidated. In a new study, a research team from Inserm, the University of Lille, and Lille University Hospital has revealed for the first time the role of tanycytes in the equation. The dysfunction of these cells, already known to ensure certain exchanges between the blood system and the cerebrospinal fluid circulating inside the brain, could be responsible for the abnormal accumulation of Tau. After demonstrating the involvement of tanycytes in the transport of the Tau protein in animals and humans, the researchers discovered that the structure of tanycytes was degraded in the brains of patients who had died from the disease. These findings are published in the journal Cell Press Blue[1].
Alzheimer’s disease manifests itself through progressive memory impairment, executive function[2] decline, and impaired temporal and spatial orientation. It is caused by a slow and progressive degeneration of neurons in the hippocampus before spreading to the entire brain.
The diagnosis of Alzheimer’s disease can be based on measuring the presence of a protein in the cerebrospinal fluid (CSF), called Tau. In healthy individuals, the presence of Tau in the CSF is low: it is secreted by neurons and then eliminated in the blood. However, in people with the disease, the structure of Tau changes and it can no longer fulfill its normal role within neurons. It accumulates in a pathological form, disrupting brain function. This accumulation gradually leads to the degeneration and death of neurons, causing cognitive decline.
The mechanisms leading to the pathological accumulation of the Tau protein are not fully understood and are an active area of research.
For more than 20 years, Vincent Prévot, Inserm Research Director, and his team at the Lille Neuroscience & Cognition Research Center (Inserm/University of Lille/Lille University Hospital) have been studying the specific role of certain cells called tanycytes. These cells are known to facilitate essential exchanges between the brain and the rest of the body, particularly between the bloodstream and cerebrospinal fluid. For example, they detect and transport leptin (the satiety hormone) to the brain, and it is thanks to their function that the brain regulates appetite and energy balance.
In a new study, the team investigated for the first time the potential role of these cells in the pathological context of Alzheimer’s disease. They methodically followed several steps.
The researchers first validated that tanycytes were indeed involved in Tau transport. To do this, they injected the Tau protein into the cerebrospinal fluid and observed its path using fluorescence techniques. This initial experiment allowed them to visualize how Tau was captured in the cerebrospinal fluid by tanycytes and then transported along their extensions[3] to the blood capillaries.
From this observation, they hypothesized that tanycytes capture Tau and then release it and eliminate it into the bloodstream.
To test this second idea, the researchers studied the consequences of blocking tanycyte transport by genetically expressing botulinum toxin in the cells, which prevents them from functioning. The results showed a loss of Tau clearance from the CSF to the blood.
The researchers thus demonstrated for the first time in animals that tanycytes were the main route for clearing Tau proteins from the brain into the bloodstream.
At the same time, this time using mouse models with high levels of Tau in the CSF, they also showed that by blocking the activity of tanycytes, these mice developed the symptoms of dementia characteristic of Alzheimer’s disease, and more generally of tauopathy, a disease characterized by the accumulation of abnormal forms of the Tau protein [4], at an earlier stage.
To take their research further, the scientists studied the brains of people who had died with Alzheimer’s disease. These analyses confirmed the presence of Tau proteins in tanycytes, as had been shown in animals. They also noticed that the tanycytes were damaged: their extensions were fragmented, interrupting the communication pathway between the CSF and the blood. This alteration appears to be specific to Alzheimer’s disease—the researchers did not find it in the brains of patients with other types of dementia.
“Our results show for the first time the ability of tanycytes to transport Tau protein from the cerebrospinal fluid to the blood and the importance of these cells in the pathophysiology of Alzheimer’s disease. They suggest that the degradation of these cells contributes to Alzheimer’s disease,” explains Vincent Prévot.
“Tanycytes could therefore be considered a new therapeutic target. What if the good health of these cells could ultimately prevent the development of the disease?” concludes the researcher.
This work was conducted as part of a project funded by the European Community (ERC Synergy WATCH, No. 810331) and the Foundation for Medical Research (FRM, MND202310017920).
[1] Cell Press Blue is a new general science journal published by Cell Press.
[2] Mental abilities that enable you to plan a task, adapt your behavior, or cope with new situations.
[3] Tanycytes are cells that have long extensions, somewhat like “arms,” which extend from their cell bodies to reach the blood capillaries that establish communication between the hypothalamus and the anterior pituitary gland.
[4] Tauopathies include, among others, Alzheimer’s disease, progressive supranuclear palsy, corticobasal syndrome…Les tauopathies comprennent, entre autres, la maladie d’Alzheimer, la paralysie supranucléaire progressive, le syndrome corticobasal…

 ©
© 










 © AdobeStock
© AdobeStock