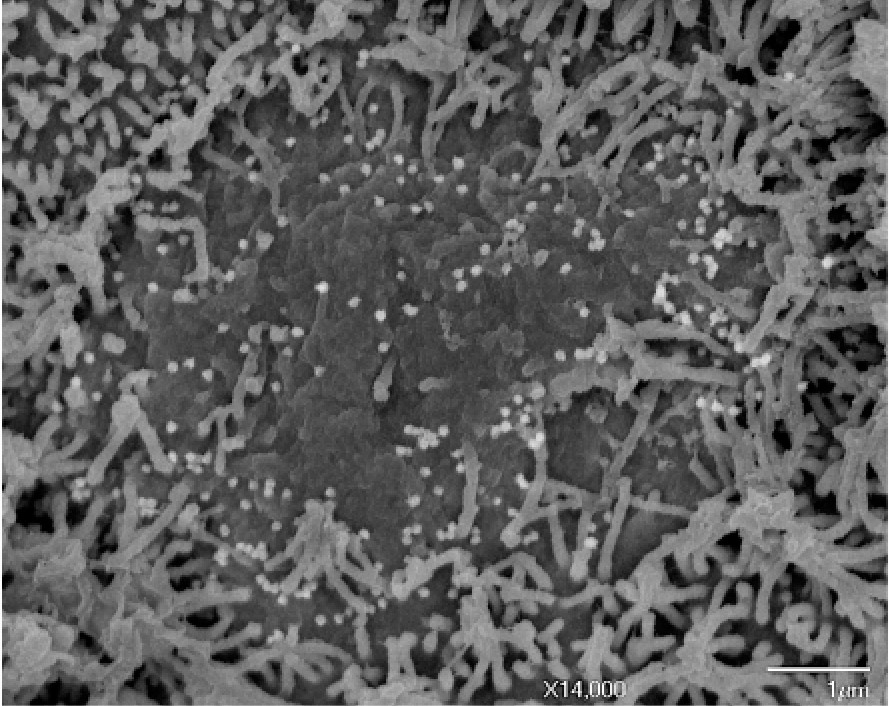
Scanning electron microscopy image showing changes in the olfactory epithelium following SARS-CoV-2 infection. The ciliated cells towards the edges of the photograph are normal. In the center, loss of cilia 2 days post-infection is discernible. Viral particles are budding off the surface of infected cells that have lost their cilia. © Perception and Memory Unit – Institut Pasteur
Loss of smell, or anosmia, is one of the earliest and most commonly reported symptoms of COVID-19. But the mechanisms involved had yet to be clarified. Scientists from the Institut Pasteur, the CNRS, Inserm, Université de Paris and the Paris Public Hospital Network (AP-HP) determined the mechanisms involved in the loss of smell in patients infected with SARS-CoV-2 at different stages of the disease. They discovered that SARS-CoV-2 infects sensory neurons and causes persistent epithelial and olfactory nervous system inflammation. Furthermore, in some patients with persistent clinical signs, anosmia is associated with prolonged epithelial and olfactory nervous system inflammation and lasting presence of the virus in the olfactory epithelium. These findings were published in the journal Science Translational Medicine on May 3, 2021.
Although COVID-19 caused by the SARS-CoV-2 virus is principally a respiratory disease, many patients present with non-respiratory symptoms. These include a sudden loss of smell in individuals infected with SARS-CoV-2, which has been reported throughout the world since the beginning of the pandemic. Until recently, there has been uncertainty as to whether the virus plays a direct role in anosmia. According to one hypothesis generally accepted until now, it was assumed that a transient edema of the olfactory clefts inhibited airflow transporting odor molecules to the olfactory neurons (the familiar sensation of a blocked nose experienced during a common cold).
In a recent study, scientists from the Institut Pasteur, the CNRS, Inserm, Université de Paris, and the Paris Public Hospital Network (AP-HP) shed light on the mechanisms involved in COVID-19-related anosmia. The study was conducted with COVID-19 patients and supplemented with tests on an animal model. This study unexpectedly demonstrates that nasopharyngeal swabs may test negative by standard RT-qPCR even if the virus is still present at the back of the nasal cavities, in the olfactory epithelium. In light of this discovery, SARS-CoV-2 diagnosis by nasal brushing may be envisaged in addition to nasopharyngeal swabbing for the PCR test in patients experiencing loss of smell.
This work also sheds light on the mechanism of COVID-19-related smell loss by revealing a series of chronological steps:
1) Cilia carried by sensory neurons are lost post-viral infection. These cilia enable the sensory neurons to receive odor molecules;
2) Virus present in sensory neurons;
3) Disruption of the olfactory epithelium (sensory organ) integrity linked to apoptosis (i.e. cell death). The epithelium is organized into regular lamellae, which are destructured by coronavirus infection;
4) Virus dissemination to the olfactory bulb, which is the first cerebral relay station in the olfactory system;
5) Inflammation and viral RNA present in several regions of the brain.
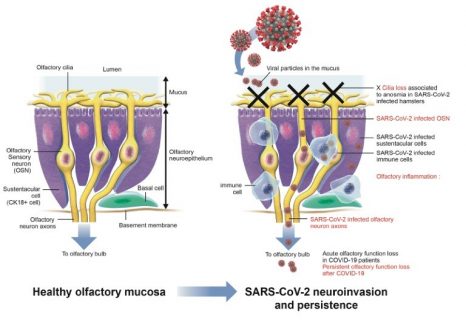
Diagram representing the various steps occurring in the sensory system and contributing to COVID-19-related anosmia.
This study demonstrates that loss of smell is also caused by deterioration of the sensory organ at the back of the nasal cavities. “We observed that SARS-CoV-2 infects not only the sensory neurons, but also the olfactory nerve and the olfactory nerve centers in the brain,” comments Pierre-Marie Lledo, CNRS scientist, head of the Perception and Memory Unit (Institut Pasteur/CNRS), and co-author of the study.
“Another key finding from this study emerged from an observation of animal models, which revealed that once the virus enters the olfactory bulb, it spreads to other nerve structures, where it induces a major inflammatory response,” explains Hervé Bourhy, head of the Lyssavirus Epidemiology and Neuropathology Unit at the Institut Pasteur and co-author of the study. Infection of the olfactory neurons may therefore provide a gateway to the brain and explain why some patients develop various psychological clinical signs (anxiety disorders, depression) or those of a neurological nature (cognitive decline, susceptibility to developing a neurodegenerative disease), for which further studies are necessary.
Marc Lecuit, head of the Biology of Infection Unit (Institut Pasteur, Inserm, Université de Paris, AP-HP) and co-author of the study concludes: “According to our results, loss of smell in COVID-19 may persists for several months in some patients and this persistence of clinical signs may be attributed to the persistence of the virus and inflammation in the olfactory mucosa.” These observations should be used to adapt the diagnosis and management of long-term COVID-19 signs.
In summary, this study has led to the following 4 key findings:
- The virus can be detected by nasal brushing in instances where it is not detected by swabs;
- SARS-CoV-2 may persist in the olfactory epithelium for several months;
- SARS-CoV-2 infects sensory neurons and prompts immune cell recruitment in the sensory organ;
- SARS-CoV-2 may cause persistent inflammation of the olfactory epithelium and the olfactory nervous system.
These contents could be interesting :
COVID-19-associated olfactory dysfunction reveals SARS-CoV-2 neuroinvasion and persistence in the olfactory system
Science Translational Medicine, 3 mai 2021
Guilherme Dias de Melo1‡, Françoise Lazarini2‡, Sylvain Levallois3‡, Charlotte Hautefort4‡, Vincent Michel2,5, Florence Larrous1, Benjamin Verillaud4, Caroline Aparicio6, Sebastien Wagner2, Gilles Gheusi2, Lauriane Kergoat1, Etienne Kornobis7,8, Flora Donati9,10, Thomas Cokelaer7,8, Rémi Hervochon11, Yoann Madec12, Emmanuel Roze13, Dominique Salmon14, Hervé Bourhy1*, Marc Lecuit3,15*, Pierre-Marie Lledo2*†
1 Lyssavirus Epidemiology and Neuropathology Unit, Institut Pasteur, 75015 Paris, France.
2 Perception and Memory Unit, Institut Pasteur and CNRS UMR3571, 75015 Paris, France.
3 Biology of Infection Unit, Institut Pasteur, Paris, France; Inserm U1117, 75015 Paris, France.
4 Otolaryngology-head and Neck Surgery Department, Hopital Lariboisiere, Assistance Publique – Hôpitaux de Paris, Inserm U1141, Université de Paris, 75010 Paris, France.
5 Institut de l’Audition, Institut Pasteur, Paris, France ; Inserm U1120, 75012 Paris, France
6 Emergency Department, Hôpital Lariboisière, Assistance Publique – Hôpitaux de Paris, Université de Paris, 75010 Paris, France.
7 Plateforme Technologique Biomics – Centre de Ressources et Recherches Technologiques (C2RT), Institut Pasteur, 75015 Paris, France.
8 Hub de Bioinformatique et Biostatistique – Département Biologie Computationnelle, Institut Pasteur, USR 3756 CNRS, 75015 Paris, France.
9 National Reference Center for Respiratory Viruses, Institut Pasteur, 75015 Paris, France.
10 Molecular Genetics of RNA Viruses, CNRS UMR3569, Université de Paris, Institut Pasteur, 75015 Paris, France.
11 Otolaryngology-head and Neck Surgery Department, GHU Pitié-Salpêtrière, Assistance Publique-Hôpitaux de Paris, Sorbonne Université, 75013 Paris, France.
12 Emerging Diseases Epidemiology Unit, Institut Pasteur, 75015 Paris, France.
13 Sorbonne Université, AP-HP, Hôpital de la Pitié-Salpêtrière, Département de Neurologie, Inserm U1127, CNRS UMR 7225, Institut du Cerveau, 75013 Paris, France.
14 Infectious Diseases and Immunology Department, Cochin Hotel Dieu Hospital, Assistance Publique – Hôpitaux de Paris, Université de Paris, 75015 Paris, France.
15 Université de Paris, Necker-Enfants Malades University Hospital, Division of Infectious Diseases and Tropical Medicine, Institut Imagine, AP-HP, 75015 Paris, France.
‡ These authors contributed equally
* These authors share senior authorship
† Corresponding author