Researcher Contact
Ali Amara
U944 Génome, Biologie Cellulaire et Thérapeutique
Institut de Recherche Saint-Louis
Vassili Soumelis
U976 Immunologie humaine, physiopathologie et immunothérapie – HIPI
Institut de Recherche Saint-Louis
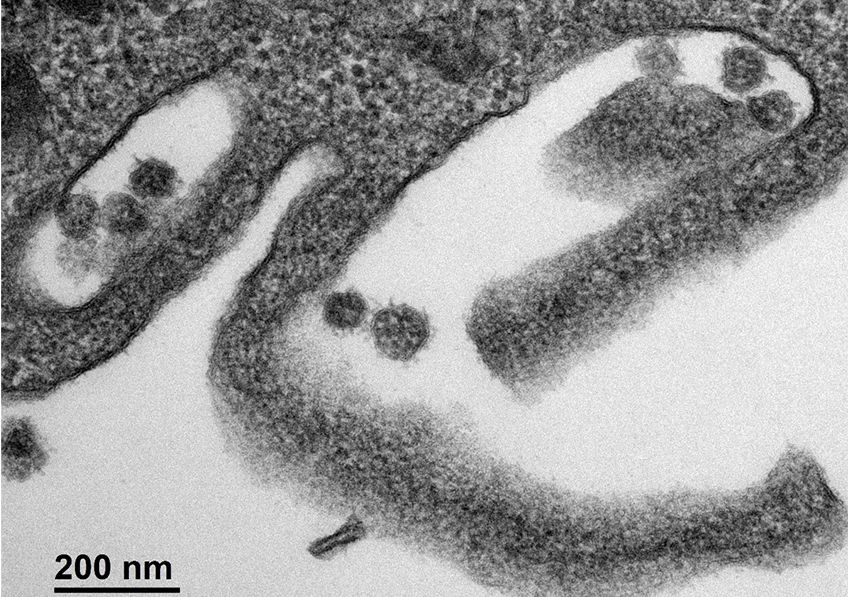
Cellule infectée par le SARS-CoV-2. © Sébastien Eymieux et Philippe Roingeard, INSERM – Université de Tours
As the COVID-19 pandemic continues, scientists are making significant headway in understanding the transmission of the SARS-CoV-2 coronavirus and the immune response it triggers at the time of infection. Researchers from Inserm, the Paris hospitals group AP-HP and Université de Paris, in collaboration with Rockefeller University in New York, have provided new data on the very early stages of immune response. Their findings have been published in Journal of Experimental Medicine.
Understanding the immune response against SARS-CoV-2 is essential if we are to know who is at risk of developing severe forms of COVID-19 and how to treat them effectively. While many studies have been conducted in patients in the advanced stages of infection, when they are already showing signs of severity, little is known about the very early stages of immune response against the virus.
Thanks to close collaboration between the Inserm teams of Ali Amara, virologist, and Vassili Soumelis, immunologist at Saint-Louis Research Institute (Université de Paris/Inserm/AP-HP), a study published in the Journal of Experimental Medicine was able to characterize innate immune response[1] within 24 to 48 hours following contact with the SARS-CoV-2 virus.
The researchers used immune cells called “plasmacytoid predendritic cells” as a model of innate immune cells that play an essential role in antiviral immunity by producing large amounts of interferon-alpha[2].
Analysis of this in vitro reconstituted response showed that SARS-CoV-2 induced effective and complete activation of the plasmacytoid predendritic cells. These then produced large amounts of interferon-alpha (the first line of defense against viruses) and differentiated into dendritic cells capable of activating T cells (which correspond to specific immunity cells). The researchers were also able to show that this activation of the plasmacytoid predendritic cells was partially inhibited by hydroxychloroquine, which would call for caution in the use of this molecule.
In the second part of the study, the teams collaborated with Jean-Laurent Casanova’s team from the Imagine Institute (Inserm/Université de Paris/AP-HP) and Rockefeller University in New York, in order to study the response of plasmacytoid predendritic cells from patients with genetic deficits for certain important genes of innate immunity. The objective was to clarify the molecular mechanisms involved in the response of these immune cells to SARS-CoV-2.
These experiments performed on samples obtained directly from patients showed that the response of plasmacytoid predendritic cells is dependent on UNC93B and IRAK-4, two important molecules of innate antiviral immunity. This research as a whole makes it possible to clarify early immune response to SARS-CoV-2 as well as some of its molecular determinants.
[1] Innate immunity is the body’s first line of defense and is triggered as soon as the body is exposed to a bacterium or virus (such as SARS-CoV-2). The innate immunity cells can contribute to the total destruction of the detected microbes or present them to the mechanisms of acquired immunity to facilitate their destruction by specific mechanisms (T and B cells).
[2] Interferons are cytokines (proteins) whose production is induced following viral, bacterial or parasitic infection, or the presence of tumor cells. While their main function is to interfere with viral replication, they also have an antibacterial and antiproliferative action, as well as an activating effect on other immune cells.
Ali Amara
U944 Génome, Biologie Cellulaire et Thérapeutique
Institut de Recherche Saint-Louis
Vassili Soumelis
U976 Immunologie humaine, physiopathologie et immunothérapie – HIPI
Institut de Recherche Saint-Louis
SARS-CoV-2 induces human plasmacytoid pre-dendritic cell diversification via UNC93B and IRAK4
Fanny Onodi1, Lucie Bonnet-Madin2, Laurent Meertens2, Léa Karpf1, Justine Poirot1,
Shen-Ying Zhang3, 4, 5, Capucine Picard4, 6, 8, Anne Puel3, 4, 5, Emmanuelle Jouanguy3,
4, 5, Qian Zhang5, Jérôme Le Goff 1, 7, Jean-Michel Molina2, 7, Constance Delaugerre2,
7, Jean-Laurent Casanova3, 4, 5, 8, 9, Ali Amara2, # and Vassili Soumelis1, 10
1 Université de Paris, Institut de Recherche Saint-Louis, INSERM U976, Hôpital
Saint-Louis, 75010 Paris, France
2 Université de Paris, Institut de Recherche Saint-Louis, INSERM U944 CNRS 7212,
Hôpital Saint-Louis, 75010 Paris, France
3 Laboratory of Human Genetics of Infectious Diseases, Necker Branch, INSERM,
Necker Hospital for Sick Children, Paris, France, EU
4 Université de Paris; INSERM UMR 1163 Institut Imagine, France EU
5 St. Giles Laboratory of Human Genetics of Infectious Diseases, Rockefeller Branch,
The Rockefeller University, New York, NY, USA
6 Study center for primary immunodeficiencies, Necker Hospital for Sick Children
Assistance Publique-Hôpitaux (AP-HP) de Paris, Paris, France, EU
7 Laboratoire de Virologie et Département des Maladies Infectieuses, Hôpital Saint-
Louis, APHP, 75010 Paris, France
8 Pediatric Hematology and Immunology Unit, Necker Hospital for Sick Children, APHP,
Paris, France, EU
9 Howard Hughes Medical Institute, New York, NY, USA
10 Assistance Publique-Hôpitaux de Paris (AP-HP), Hôpital Saint-Louis, Laboratoire
d’Immunologie, F-75010, Paris, France