The team led by Hélène Puccio, director of research for Inserm at the Institute of Genetics and Molecular and Cellular Biology (IGBMC) (Inserm / CNRS / University of Strasbourg) in close collaboration with Patrick Aubourg’s team (Inserm and Professor of Neuropaediatrics at Bicêtre Hospital) has demonstrated, in the mice, the efficacy of gene therapy for treating the heart disease associated with Friedreich’s ataxia, a rare hereditary neuro-degenerative disorder. The transfer, via a viral vector, of a normal copy of the gene deficient in patients, allowed to fully and very rapidly cure the heart disease in mice. These findings are published in Nature Medicine on 6 April, 2014.
Friedreich’s ataxia is a severe, rare hereditary disorder which combines progressive neuro-degeneration, impaired heart function and an increased risk of diabetes. The condition affects one in every 50,000 birth. There is currently no effective treatment for this disease. In most cases, Friedreich’s ataxia starts in adolescence with impaired balance and coordination (ataxia) of voluntary arm and leg movements, confining the majority of patients to a wheelchair after 10-20 years’ progression. However, complications affecting the heart are the major cause of death in 60% of patients, most often before the 35 years of age.
The disease is caused by a common mutation in the FXN gene which leads to a dramatic decrease in the production of the protein named ‘frataxin’. The reduced frataxin level disturbs the activity of mitochondria. These organelles are essential to cells and play a fundamental role in energy production. The nerve tissue (cerebellum, spinal cord etc.) and heart tissue are particularly vulnerable to this shortage of energy, which can lead to fatal heart failure.
The teams led by Hélène Puccio, director of research at Inserm and Patrick Aubourg have developed a therapeutic approach based on the use of an adeno-associated virus (AAV)[1], which is known to efficiently target and express a therapeutic gene in heart cells. The virus was modified to make it harmless but nevertheless capable of introducing a normal copy of the FXN gene in the heart cells, thus leading to the expression of frataxin.
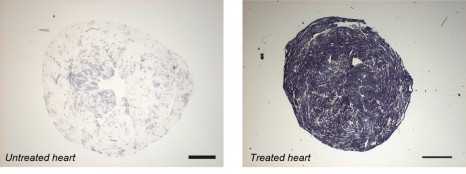
The illustrations show a measurement of the activity of a mitochondrial protein (in blue) essential for cellular energy production, which is impaired when frataxin is absent (no staining in the untreated heart). Using gene therapy expressing frataxin, the activity of this essential protein can be corrected across the heart’s entire surface. © Inserm / H. Puccio