Researcher Contact
Benoît Gamain Directeur de recherche CNRS UMR-S 1134, Inserm/Université de Paris Tel : +33 (0)1 44 49 31 47 E.mail: rf.mresni@niamag.tioneb

©Benoît Gamain. Gestational malaria is associated with low birth weight for the baby and an over-risk of neonatal mortality.
Malaria infection during pregnancy represents a major public health problem in the regions endemic for the disease, substantially increasing the risks to mothers and their unborn children. For newborns, malaria is linked to low birth weight and an excess risk of mortality. To protect this population, a team of researchers from Inserm and Université de Paris led by CNRS Research Director Benoît Gamain is developing a vaccine at the French National Institute of Blood Transfusion (INTS). Called PRIMVAC, the vaccine has undergone a clinical trial to study its safety and collect preliminary data on its ability to induce an immune response. The results of this clinical trial sponsored by Inserm[1] have been published in the prestigious journal Lancet Infectious Diseases.
In the areas of the world where malaria is endemic, people acquire immunity throughout their childhood, meaning that they are generally protected against its most severe outcomes once they reach adulthood. However, pregnant women are an exception because the red blood cells infected with the Plasmodium falciparum parasite responsible for malaria accumulate in the placenta, promoting anemia and gestational hypertension. The disease is also linked to a higher risk of spontaneous abortion, premature birth and intrauterine growth delays which lead to low birth weight and a high rate of neonatal mortality. In Sub-Saharan Africa, 11 million pregnant women were infected with malaria in 2018, with around 900,000 of their babies born underweight.
To tackle this public health problem, a team of researchers from Inserm and Université de Paris led by CNRS Research Director Benoît Gamain has spent the past two decades developing a vaccine for gestational malaria. The goal is to prevent the deaths of up to 10,000 mothers and 200,000 babies each year. “Developing an effective vaccine for young women before their first pregnancy is a priority if we are to reduce malaria-related mortality. An effective strategy could focus on a population similar to that targeted by HPV vaccination, for example, before the women become sexually active”, emphasizes Benoît Gamain.
A safe and effective vaccine
Called PRIMVAC, the vaccine had recently been produced in large quantities in accordance with current regulations. In a clinical trial published in Lancet Infectious Diseases, the researchers provide data on its safety and ability to induce an appropriate immune response, up to 15 months after the initial vaccination.
The vaccine was evaluated in 68 non-pregnant women aged 18 to 35 at the Cochin Pasteur Clinical Investigation Center in Paris, then at the National Center for Research and Training on Malaria (CNRFP) in Ouagadougou, Burkina Faso. The participants were randomly assigned to 4 cohorts, receiving the vaccine at various doses, on 3 occasions over a period of 3 months. These women were then monitored for 15 months in order to identify and treat any side effects and study the immune response induced by the vaccination.
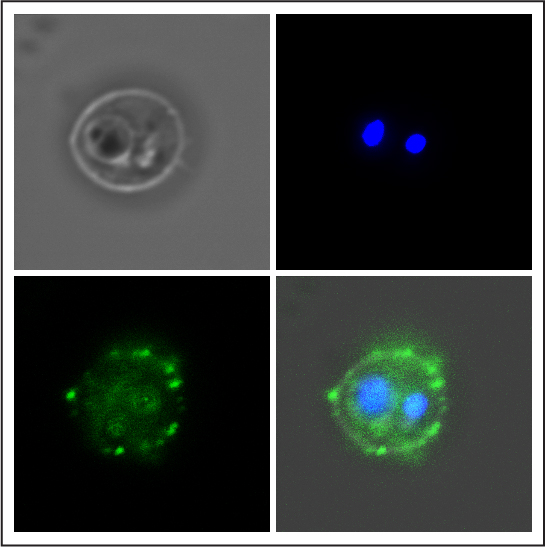
Antibody (green) of a vaccinated volunteer binding to the surface of a human red blood cell infected with the Plasmodium falciparum parasite (blue). Credits: Inserm/Chêne, Arnaud et Semblat, Jean-Philippe
“We were able to show that the vaccine is well tolerated, at all the tested doses. The side effects observed were mainly pain at the injection site. We also revealed that the quantity of antibodies generated by the vaccine increases after each vaccination and that they persist for several months. It therefore appears that the vaccine has the capacity to trigger a lasting and potentially protective immune response”, underlines Gamain.
Studying this immune response on the longer term will be the subject of future clinical trials. The researchers want to continue monitoring the 50 Burkinabe volunteers in order to evaluate whether the immune response induced by the vaccination is maintained until their first pregnancy.
[1] The trial was coordinated by the Cochin Pasteur Clinical Investigation Center in Paris and the EUCLID/F-CRIN clinical trials platform in Bordeaux in collaboration with the National Center for Research and Training on Malaria (CNRFP) in Ouagadougou, Burkina Faso, and the European Vaccine Initiative (EVI). Funding: Federal Ministry of Education and Research, through the development bank KfW, Germany; Inserm, and National Institute of Blood Transfusion (INTS), France; Irish Aid, Department of Foreign Affairs and Trade, Ireland.
Benoît Gamain Directeur de recherche CNRS UMR-S 1134, Inserm/Université de Paris Tel : +33 (0)1 44 49 31 47 E.mail: rf.mresni@niamag.tioneb
Sources PRIMVAC vaccine adjuvanted with Alhydrogel or GLA-SE to prevent placental malaria: a first-in-human, randomised, double-blind, placebo-controlled study Sodiomon B. Sirima1,2, Laura Richert3,4,5, Arnaud Chêne6,7,8, Amadou T. Konate1, Cécilia Campion3, Sébastien Dechavanne6,7,8, Jean-Philippe Semblat6,7,8, Nadine Benhamouda9, Mathilde Bahuaud10, Pierre Loulergue11, Alphonse Ouédraogo1, Issa Nébié1,2, Moïse Kabore2, Désiré Kargougou1, Aïssata Barry1, San Maurice Ouattara1, Valérie Boilet3, Florence Allais3, Gwenaelle Roguet11, Nicolas Havelange12, Elvira Lopez-Perez13, Alexis Kuppers13, Eleine Konaté11, Caroline Roussillon3,14, Myriam Kanté3, Linda Belarbi11, Amidou Diarra1,2, Noelie Henry1, Issiaka Soulama1, Amidou Ouédraogo1, Helene Esperou13, Odile Leroy12, Frederic Batteux10, Eric Tartour9, Nicola K. Viebig12, Rodolphe Thiebaut3,4,5*, Odile Launay11*, Benoît Gamain6,7,8 1 Centre national de recherche et de formation sur le paludisme, 01 P.O. Box 2208, Ouagadougou 01, Burkina Faso. 2 Groupe de Recherche Action en Santé (GRAS) 06 BP 10248 Ouagadougou 06, Burkina Faso. 3 Univ. Bordeaux, Inserm, Bordeaux Population Health Research Center, UMR 1219; CHU Bordeaux; CIC 1401, EUCLID/F-CRIN Clinical Trials Platform, F-33000, Bordeaux, France. 4 CHU Bordeaux ; Pôle de Santé Publique, F-33000, Bordeaux, France. 5 Inria SISTM team, F-33405, Talence, France. 6 Université de Paris, Biologie Intégrée du Globule Rouge, UMR_S1134, BIGR, INSERM, F-23 75015, Paris, France. 7 Institut National de la Transfusion Sanguine, F-75015, Paris, France. 8 Laboratory of excellence GR-Ex, F-75015, Paris, France. 9 INSERM U970, Paris Cardiovascular Research Centre, Université de Paris, Faculté de médecine, Hôpital Européen Georges Pompidou, Service d’Immunologie Biologique, F-75015, Paris, France. 10 Assistance Publique Hôpitaux de Paris (AP-HP), Hôpital Cochin, Plateforme d’immuno-monitoring vaccinal, Laboratoire d’Immunologie, F-75679, Paris, France. 11 Université de Paris, Faculté de médecine, INSERM, CIC 1417; F-CRIN I-REIVAC; AP-HP, hôpital Cochin, CIC Cochin Pasteur, F-75679 Paris, France. 12 European Vaccine Initiative, UniversitätsKlinikum Heidelberg, 69115 Heidelberg, Germany. 13 INSERM, Institut de Santé Publique, Pôle de Recherche Clinique, F-75013, Paris, France. 14 Direction de la Recherche Clinique et de l’Innovation, Unité de sécurité et vigilance des Essais Cliniques, CHU de Bordeaux, F-33000, Bordeaux Lancet Infectious Diseases, Février 2020 DOI : https://doi.org/10.1016/S1473-3099(19)30739-X