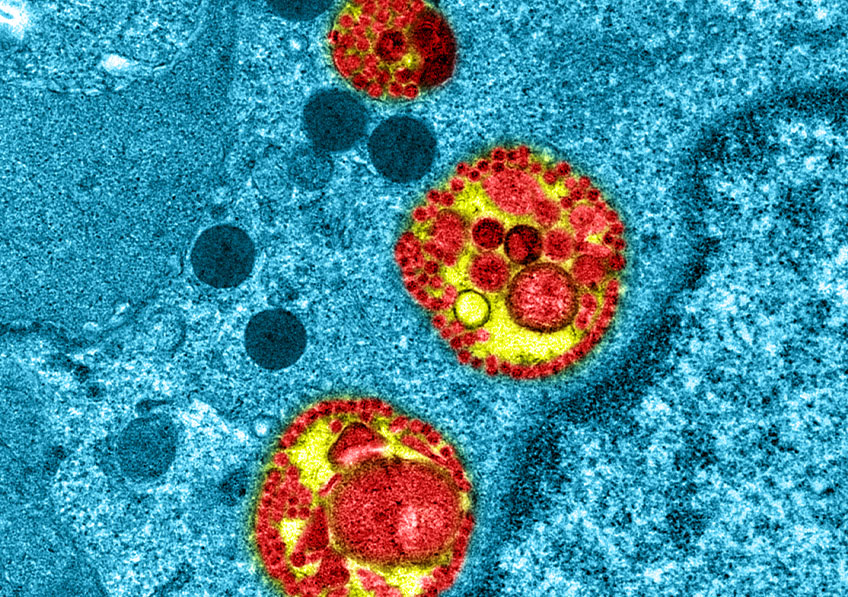
Intracellular observation of reconstituted human respiratory epithelium MucilAir™ infected with SARS-Cov-2 © Manuel Rosa-Calatrava, Inserm ; Olivier Terrier, CNRS ; Andrés Pizzorno, Signia Therapeutics ; Elisabeth Errazuriz-Cerda UCBL1 CIQLE. VirPath (Centre International de Recherche en Infectiologie U1111 Inserm – UMR 5308 CNRS – ENS Lyon – UCBL1). Colorized par Noa Rosa C.
1. React rapidly to accelerate research and tackle the pandemic
First identified in the central Chinese city of Wuhan in December 2019, the novel SARS-CoV-2 coronavirus has rapidly spread across the world, leading to the most serious pandemic in recent history. Inserm, as a leading biomedical research stakeholder in France and internationally, has been mobilized from the very beginning of this global health crisis, thanks to the commitment of its many experts in subjects related to fundamental research, therapeutic research, and modelling. Participating also in the French solidarity effort, the Institute has distributed tens of thousands of masks, gloves, gowns, shoe covers and reagents to medical teams working in the nation’s hospitals.
By May 6, 2020, Inserm had already been involved in 167 scientific publications on COVID-19 (including 19 preprints), reflecting the unfailing energy and proactiveness of its researchers. This research primarily concerns potential therapeutic approaches, the search for a vaccine, epidemiology, the deployment of telemedicine, predicting the spread of the virus in various countries, and its transmission. A large part of the research published has received REACTing consortium seed funding.
A key player in fighting the COVID-19 pandemic, REACTing – which stands for REsearch and ACTion targeting emerging infectious diseases – launched and coordinated by Inserm since 2013 under the egis of Aviesan, has been tasked with preparing and coordinating French research into emerging infectious diseases in order to prevent and fight epidemics.
A number of task forces (New Therapeutic Approaches, COVID-19 Vaccines, Animal Models, Modelling, and Digital) have been created in order to reflect on the major research priorities and evaluate the projects submitted to the consortium by research teams from across France and internationally.
In addition, Inserm is involved in various World Health Organization (WHO) working groups, the Scientific Advisory Board consulted by the French government, and the Analysis, Research and Expertise Committee (CARE) set up by the French Ministry of Solidarity and Health.
Never has Inserm’s mission, Science for Health, been so necessary. Although many questions remain unanswered concerning the evolution of the pandemic, Inserm will continue its efforts to inform public decision-making with research of excellence that combines rigor and ethics.
2. Treat patients
Within REACTing, the New Therapeutic Approaches Task Force meets weekly to evaluate the many projects submitted to it regarding research into treatment avenues. Experts in the various themes addressed are invited in order to enrich discussions, in addition to members of CARE, the French Health Directorate, MESRI, and the REACTing COVID-19 Scientific Advisory Board.
The antivirals approach: focus on the Discovery trial
Discovery is a European project whose French component has begun thanks to REACTing seed funding paid jointly by MESRI and the Ministry of Solidarity and Health. It is also funded by the Program for Clinical Research in Hospitals (PHRC) and has been incorporated in the WHO international Solidarity trial.
The trial evaluates the efficacy of various antiviral treatments in limiting the viral multiplication observed in certain hospitalized patients whose condition often deteriorates on around day seven of the disease.
It intends to recruit 3,200 European patients with moderate to severe COVID-19, at least 800 of whom in France, admitted to a medical department or directly to intensive care. Over 750 patients have been enrolled in Discovery so far, mainly in France.
- The treatments
The objective of Discovery is to evaluate the efficacy and safety of various experimental therapeutic strategies which, according to current scientific knowledge, have been identified as potential therapeutic candidates for COVID-19. To recap, it involves testing and comparing the following five strategies:
- optimal standard of care;
- optimal standard of care plus remdesivir;
- optimal standard of care plus lopinavir and ritonavir;
- optimal standard of care plus lopinavir, ritonavir and interferon beta;
- optimal standard of care plus hydroxychloroquine.
Therefore, none of the patients enrolled in the trial is left without treatment and none of the patients is receiving placebo.
- Why an open-label randomized trial?
Discovery is a randomized trial, meaning that the treatment is not chosen by the doctor but assigned randomly. Each arm of the clinical trial is assigned an equal number of patients so that it is balanced, with sufficient data obtained for each treatment tested.
An open-label trial design was chosen in order to save valuable time during this pandemic. It must be remembered that the molecules tested are not all available in the same dosage forms. For a double-blind trial to be possible, it would take a long time to prepare placebos that resemble each of the treatments being tested, therefore delaying the start of the trial.
However, in order to limit bias, although the patients and doctors know which treatment is being administered, the researchers responsible for statistical analysis do not. This controlled, open-label trial design will therefore enable results to be obtained as rapidly as possible whilst respecting rigorous and high-quality methodology.
The immunomodulation approach: focus on Corimuno-19
Some hospitalized patients present a specific profile in which the deterioration in their condition seems to be due to their excessive immune response to the infection rather than to the multiplication of the virus. One avenue currently explored by Inserm researchers is to understand and evaluate the effect on COVID-19 patients of treatments that would modulate this response.
This is the challenge of the Corimuno-19 project, a cohort of open-label, randomized, controlled trials. The overall objective of this large-scale study is to test various treatments (especially immunomodulator treatments) and determine which present the most favorable risk/benefit ratio in adult patients hospitalized for COVID-19 pneumonia – diagnosed either at the moderate to severe stage and requiring no mechanical ventilation, or at the critical stage requiring mechanical ventilation.
The antibodies approach: focus on Coriplasm
Particularly mediatized is Coriplasm – one of the Corimuno-19 clinical trials, and which is sponsored by the Paris hospitals group (AP-HP). With the support of Inserm and REACTing, the French Blood Establishment (EFS) is deploying a process to enable the collection, qualification, preparation and provision to the clinical teams of plasma from convalescent patients. The idea is to evaluate whether their plasma is capable of immediately transferring this immunity to other patients, as had been the case when treating various infectious respiratory diseases such as SARS-CoV-1, MERS-CoV or H1N1 influenza. The objective is to determine whether this strategy reduces the frequency of severe forms of COVID-19 and their associated mortality.
Other therapeutic research initiatives
Other drug repurposing strategies
Therapeutic repurposing consists of finding new therapeutic indications for drugs that are well-known, safe, and already available in the pharmacopeia. Within the context of the COVID-19 pandemic, a number of teams are testing molecules used for other diseases on SARS-CoV-2, in order to evaluate their potential antiviral or immunomodulating effects. The team of Inserm Research Director Manuel Rosa-Calatrava at the International Center for Research in Infectious Diseases (Inserm/Université Claude-Bernard Lyon 1/CNRS/ENS Lyon) is working on the implementation of a strategy to repurpose drugs for new antiviral therapeutic indications. An approach that his group had already validated by repurposing an antihypertensive drug as an inhibitor of the influenza viruses (a phase 2 clinical trial has been ongoing over the past three winters). His team had already repurposed two other commercially available molecules in vitro against MERS-CoV. The researchers are now testing these molecules on cell lines and in their unique ex vivo model of reconstituted human respiratory epithelium infected with SARS-CoV-2.
The Corona accelerated R&D in Europe (CARE) program, funded by the Innovative Medicines Initiative, is led by the Vaccine Research Institute under the supervision of Inserm in partnership with 36 research teams from a number of European countries, China, and the US. It has two key objectives: urgently develop effective molecules for the current COVID outbreak and find treatments that could be useful in controlling future epidemics, should other coronaviruses emerge. The scientists have developed an ambitious research and development program in order to facilitate the identification of candidate drugs for therapeutic repurposing. The project will also include a component focused on developing new molecules. All relevant molecules will be tested in vitro and on laboratory animal models, following which the most promising candidates will undergo large-scale clinical trials.
For more information: https://www.imi.europa.eu/news-events/press-releases/imi-announces- covid-projects-boosts-funding-pot-eur-72-million
MacCOV: a project to combat acute respiratory distress
In Villejuif, the team of Jean-Luc Perfettini (Inserm/Université Paris-Saclay/Gustave Roussy) is working on a new COVID-19 therapeutic strategy that involves reprogramming certain immune system cells (macrophages) in order to reduce their ability to promote inflammation.
Data available on the disease indicate that one of its severe complications, acute respiratory syndrome, is due to lung tissue lesions caused by a massive influx of inflammatory molecules (cytokines), which the specialists are even referring to as a “cytokine storm”. However, given that these pro-inflammatory molecules are essentially produced by the macrophages, the researchers hope that reprogramming these immune cells will prevent cytokine storm.
Corona-Pep-Stop: a project to develop fusion inhibitory peptides
The aim of the Corona-Pep-Stop project, led by Inserm Research Director Branka Horvat at the International Center for Research in Infectious Diseases (Inserm/Université Claude Bernard Lyon 1/CNRS/ENS Lyon), is to transpose findings from the team’s previous research into the measles and Nipah viruses. Their strategy is to block the entry of the viruses into cells using specific peptides of the viral fusion protein.
This protein enables viruses with a membrane, such as SARS-CoV-2, to fuse their membrane with that of the host cells in order to transfer their genome. The researchers hope to identify a new class of antiretroviral drugs (fusion inhibitory peptides) that target this protein and are effective against several coronaviruses. These new molecules could then be used for the prevention and treatment of SARS-CoV-2, and also against other coronaviruses that may emerge in the future.
CoV2-E-TARGET: venom vs. virus
Led notably by Inserm researcher Michel De Waard at the Thorax Institute (Inserm/Université de Nantes/CNRS), the aim of this project is to identify a treatment that targets a protein of the viral envelope – protein E – which has an ion channel function (in that it enables the selective passage of certain ions through the cell membrane and facilitates the entry of the virus into the cells). The laboratory has at its disposal a collection of different types of venom that includes over 40,000 peptides, a large number of which target the ion channels in very specific ways. The idea is to identify potential peptides derived from animal venom that could block the passage of the ions and prevent the replication of the virus in the host cells.
3. Find an effective vaccine
Although phase I clinical trials to test candidate vaccines have now been launched in the USA and China, many questions persist concerning immune response to the virus and a potential vaccine.
Throughout the world, over one hundred teams are working on the development of vaccines, including a number of French groups. Of the thirty or so teams working on the candidate vaccines in France, twelve are from Inserm. Although their development work remains in the early stages for the most part, it is still furthering knowledge of the virus and vaccine research as a whole.
In order to provide support and identify research priorities, REACTing has set up a dedicated COVID-19 Vaccines Task Force. Its role is to collect information on the progress of the various candidate vaccines, catalogue the research of the French teams, and discuss the most relevant measures to deploy in the national context.
This Task Force has defined criteria for assigning priority to the French teams’ COVID-19 candidate vaccine development initiatives, which include the speed of the vaccine production cycle, minimization of the risk of disease exacerbation caused by immune mechanisms, the potential for producing the vaccine on a very large scale, and the availability of preclinical test results suggestive of the induction of disease protection.
The search for a new vaccine
REACTing COVID-19 Vaccines Task Force member Frédéric Tangy (Institut Pasteur) leads a team that is working on a COVID-19 candidate vaccine at quite an advanced stage of development. It uses as a platform the attenuated measles vaccine that had already been used in the development of certain candidate vaccines, notably against Chikungunya. A phase I clinical trial is scheduled for September 2020.
The Vaccine Research Institute also has a role to play in vaccines research within the context of the COVID-19 pandemic. Its researchers are mobilized for the accelerated development of an SARS-CoV-2 coronavirus vaccine based on its expertise and technology developed for other infectious diseases, such as HIV. The novel approach of this Institute is based on the targeting of dendritic cells, key cells in immune response.
Thanks to the involvement of this institute in French Covid-19 – the national cohort of patients infected with SARS-CoV-2, coordinated by REACTing in line with 56 hospitals in France, the objective of the research is to characterize patient immune response. Understanding this aspect of the infection is an essential prerequisite for the development of any vaccine.
The BCG vaccine to protect medical staff?
Several studies suggest that some live vaccines, such as BCG or the oral polio vaccine, have non-specific beneficial effects on certain infections. Thereforeit is possible that BCG could reduce the intensity of SARS-CoV-2 infection by stimulating the memory of innate immunity, the first line of immunity in the face of infection, and thereby induce “trained innate immunity”. Furthermore, what few contraindications there are to the use of this vaccine are well known, and its very low cost is an advantage.
An Inserm team is preparing the implementation of a French double-blind trial to test the non-specific protective effects of the BCG vaccine, the idea being to evaluate whether it could offer medical staff a certain level of protection against COVID-19. Collaboration between this team and Spanish scientists who are also conducting research in the area would enable large-scale comparison of the benefits of BCG versus a placebo common to both countries. Should such a trial go ahead, the participants would need to be followed up for several months in order to obtain reliable data.
4. Understand the disease better
French Covid-19
French Covid-19 is an observational french cohort launched at the end of January that has recruited over 2,000 patients hospitalized in France. It is coordinated by Yazdan Yazdanpanah, infectious diseases specialist at Bichat Hospital and Director of the Inserm Immunology, Inflammation, Infectiology and Microbiology Theme-Based Institute. Its objectives are to describe the clinical characteristics of the forms of COVID-19 that require hospitalization and to identify the factors predictive of severity (ICU admission or death). This involves studying the various clinical, biological, virologic, and imaging data available in the records of the patients who will be followed up for six months after their discharge from hospital. The findings of this research will be crucial in better equipping doctors to treat the disease.
Another study, CritiSARS2, aims to identify viral markers, inflammatory markers, and/or markers that reveal immunization in the critical forms of COVID-19. It is led by Karine Faure, head of the Department of Infectious and Tropical Diseases at Lille Teaching Hospital and researcher at the Center of Infection and Immunity of Lille (Inserm/CNRS/Université de Lille/Institut Pasteur de Lille).
Studying the risk of reinfection
Simon Fillatreau (unit 1151, Inserm/Institut Necker-Enfants malades) and his colleagues are working on a key question: are we protected from a second infection after having recovered from the first? If so, how long does this protection last and is it affected by age or any other individual parameters? A number of components carry the memory of the immune system and are likely to protect us from SARS-CoV-2 reinfection. The team is particularly interested in the memory B-cells and T Follicular Helper cells.
Following reinfection after having previously been exposed to a pathogen, these lymphocytes are responsible for an extremely rapid and substantial increase in the levels of protective antibodies. In the case of hepatitis B, for example, it is the memory cells that protect the individual in the absence of persistent antibody production.
The project, entitled MEMO-CoV2, therefore looks at the immune memory carried by the B-cells and T Follicular Helper cells specific to SARS-CoV-2, in order to estimate their persistence, longevity, and origin of their formation during the response to the initial infection.
The search for biomarkers
Harmonicov, a project led by an Inserm team in Rennes, is based on a cohort of one hundred adults under the age of 65. Based on this cohort, the researchers will analyze the immune responses of people with COVID-19, comparing those of patients who have recovered with those of critical patients requiring assisted ventilation. The objective is to identify markers of the favorable or unfavorable evolution of the disease, and also to study the memory cells that produce SARS-CoV-2 antibodies.
5. Model and monitor the epidemic
The Mathematical Modelling of Infectious Risks Task Force was set up very quickly and its modelling concerns both the general population and more specific populations, such as medical staff.
Modelling the spread of the epidemic
Since January 2020, work by the team of Inserm researcher Vittoria Colizza at the Pierre Louis Institute of Epidemiology and Public Health (Inserm/Sorbonne Université) has made it possible to model the spread of the epidemic from China to Europe and Africa, with the objective being to better orient prevention policies and improve surveillance of the epidemic.
Their first publication, in the journal Eurosurveillance, concerned the risk of importing the virus into Europe. This was based on the scenario of all Chinese provinces declaring more than ten cases at the time as well as on data from January 2019 on air travel flows from these regions to Europe, produced by the OAG (a global leader in the collection of flight data). Another study published by the group mid-February in The Lancet evaluated the risk of importing the virus into Africa, with Egypt, Algeria and South Africa presented as the countries most at risk.
Another major research avenue explored by Colizza and her team in collaboration with telecommunications group Orange is the impact of lockdown on population mobility, by studying aggregated and anonymized cell phone network data. The researchers are particularly interested in spontaneous changes in mobility occurring before and during lockdown, and their impact on the evolution of the pandemic. The collected data will also be integrated into models of pandemic spread developed by the team. This is to improve predictions of how the virus will spread and identify regions at risk of becoming clusters and having their healthcare systems overwhelmed. In a report published at the start of May, the team shows that mobility had decreased by 65% across the national territory during lockdown.
The team is also modelling potential scenarios on which to base decisions concerning the lifting of lockdown. In a report published mid-April on the EPIcx laboratory website, the researchers stress the need to support all lockdown-lifting strategies with measures involving mass testing, the identification of those having been in contact with confirmed cases, and the isolation of detected cases. A report on the role of the school reopenings was also published at the start of May, showing that reopening the elementary and preschools from May 11, whether gradual or not, would probably not lead to saturation of the ICUs. It is considered that these structures would reach 65% capacity at the most.
Surveillance of the epidemic intensified with Covidnet
Established in 2012 by the Sentinelles network (Inserm/Sorbonne Université) and the French Public Health Agency, the GrippeNet.fr study is a comprehensive information resource for epidemiologists wishing to monitor the evolution of seasonal influenza. Each year, the network collects epidemiological data on influenza directly from the population, online and anonymously. GrippeNet.fr began its ninth season at the end of November 2019 and has over 7,200 participants so far, who each week declare the symptoms that they had or had not experienced since they last logged on. For better monitoring of the current epidemic, the GrippeNet.fr study has become Covidnet.fr. Based on questionnaires sent to the volunteers of the GrippeNet.fr/Covidnet.fr cohort, it is currently the only health surveillance system in France that makes it possible to study the symptoms presented by patients having not sought assistance from the healthcare system.
The NoCov project led by Inserm researcher Chiara Poletto at the Pierre Louis Institute of Epidemiology and Public Health (Inserm/Sorbonne Université) also uses data from the Sentinelles network to model the spread of the epidemic in the short term and identify the main factors of virus transmission (notably by looking at disease transmissibility and severity according to age).
Risk of hospital transmission
The research also includes studies that aim to model the risk of SARS-CoV-2 transmission in the hospital setting (nosocomial risk). One such study is being conducted by Didier Guillemot and Lulla Opatowski, researchers at the Biostatistics, Biomathematics, Pharmacoepidemiology and Infectious Diseases laboratory (Inserm/Institut Pasteur/Université de Versailles Saint-Quentin-en-Yvelines).
6. Study lockdown and its impacts
The Sapris study and the social challenges of lockdown
A vast survey based on five large, national, general-population cohorts (Constances, Étude familiale E3N-E4N, Elfe / Epipage 2, NutriNet Santé) conducted in close coordination with the leaders of those cohorts, the Sapris project (health, practices, relationships and social inequalities) looks at the epidemiological and social challenges presented by the exceptional prevention measures deployed against COVID-19. Coordinated by Nathalie Bajos, Inserm Research Director and sociologist-demographer, and Fabrice Carrat, Professor of Public Health at Sorbonne Université in collaboration with the French Public Health Agency, the study involves a multidisciplinary group of researchers from Inserm, France’s National Institute for Demographic Studies (Ined), National Center for Scientific Research (CNRS), Université Paris-Saclay, Sorbonne Université, Sorbonne Paris Nord and Université Paris Dauphine-PSL, National Health Insurance Fund (CNAM), Institut Gustave Roussy and the National Research Institute for Agriculture, Food and Environment (INRAe), covering fields as varied as epidemiology, sociology, demographics and economics.
By means of a questionnaire sent out at the start of April (completed by 115,000 participants), and repeated several times during and after lockdown, participants are asked about the specific challenges of the epidemic and the lockdown measures. The main themes studied are the incidence of COVID-19 symptoms and other health problems, the use of treatment for other health conditions or failure to seek treatment, the perception of risk to oneself and in general, the effects of the prevention measures on daily life, social relationships, work,and the education of children. In addition, in collaboration with the Emerging Viruses Unit (Inserm/IRD) led by virologist Xavier de Lamballerie, the participants are offered serological tests that provide indications on prevalence at national level and enable study of the factors associated with SARS-CoV-2 seropositivity. These tests will be repeated in order to study the persistence of the immune response against SARS-CoV-2 and to establish correlates of protection.
In addition, the EpiCOV (Epidemiology and living conditions) project is a large-scale public health study whose objective is to provide global and representative mapping, at national and departmental level, of the immune status of the population and of its dynamic. It also aims to study living conditions in this particular context of restricted movement, taking into account the diversity of socioeconomic situations.
Under the scientific responsibility of Inserm researchers Josiane Warszawski and Nathalie Bajos, sociologist-demographer, EpiCOV is conducted in collaboration with France’s Directorate for Research, Studies, Assessment, and Statistics (DREES), National Institute of Statistics and Economic Studies (INSEE) and Public Health Agency. A sample of 200,000 people aged 15 years or over, representative of the general population, has been created by INSEE in order to provide data on a territorial level and study socioeconomic conditions – notably for those in a situation of economic precariousness. The participants are required to complete a questionnaire similar to that used in Sapris and perform a serological home test. With this project, which began in early May, it will be possible to obtain detailed information on the spatial, temporal, sociodemographic and familial aspects of the epidemic and the lockdown measures. The survey will be repeated several times in order to study the evolution and circulation of the virus and its impact on living conditions. The survey fully involves the local authorities in the initiative and the future results.
Sapris and EpiCOV both require participants to collect a drop of blood by pricking the end of their finger and pressing it four times onto the absorbent paper provided before mailing it back to the researchers in a prepaid envelope.
Evaluating the contacts of French people during lockdown
A population survey to collect and analyze data concerning the contacts between French people during lockdown has been launched by Institut Pasteur in collaboration with Inserm and several universities. Called SocialCov, this study is based on online questionnaires concerning people’s contacts before and during lockdown, with the aim of gaining deeper insight into the impact of lockdown on our social and professional lives. More specifically, the survey will make it possible to identify the number of contacts we have each day, their frequency, and distribute these results by age group.
Supporting mental health during lockdown
The measures of social distancing, and particularly the lockdown of the French population, will doubtlessly have an impact on mental health and wellbeing. Several studies have already examined this issue with, for example, a meta-analysis published in The Lancet suggesting that lockdown is associated with a harmful psychological impact, characterized by mood disorders, confusion, and in the most extreme cases by the manifestation of post-traumatic stress disorder.
For a deeper insight into these effects, Anne Giersch, Inserm researcher and head of the Cognitive Neuropsychology and Pathophysiology of Schizophrenia laboratory in Strasbourg, is conducting a study in healthy volunteers. Its objective is to explore the positive and negative effects of lockdown, particularly on mental health. The participants are asked to answer anonymous questionnaires on their general health, concern about infection risk, conditions of lockdown, social network before and during lockdown, and also on their mood, emotions and stress levels.
In order to help the general population deal with anxiety related to the pandemic, Inserm researchers have developed a COVID-19 extension of the StopBlues initiative. This initiative, based on a free website and application, was developed in 2018 by the Eceve-Inserm research team led by Karine Chevreul. Its users are assisted in order to identify the signs of their malaise, look for the possible causes and find concrete solutions for confronting it. Within the context of the pandemic, the COVID-19 extension of StopBlues includes the publication of short videos describing the emotions that can be felt in the face of fear of the disease, lockdown difficulties, social isolation and family conflicts.
Tempo Covid 19: impacts on mental health and addictive behaviors
Maria Melchior, Inserm researcher at the Pierre Louis Institute of Epidemiology and Public Health (Inserm/Sorbonne Université), and her colleagues have launched the Tempo Covid 19 project. Every week since March 24, the participants of a French cohort (Tempo) were asked questions about their health and psychological difficulties, social isolation, tobacco, alcohol and cannabis consumption, professional, financial and familial situations, and activities during the lockdown period. The aim is to evaluate the impact of the epidemic and lockdown on mental health and addictive behaviors, according to social situation. Between 400 and 500 people answered the 6 questionnaires sent to them. The researchers will survey the same participants when lockdown is lifted in order to have the most comprehensive follow-up possible.
Melchior is also working in partnership with the French Public Health Agency on Echo, a survey to evaluate knowledge of the virus and protective measures (barrier measures, lockdown) in people living in situations of exclusion and accommodated in medical-social association structures, the majority of whom are migrants. The researchers would like to survey 300 people by the end of May.
Epidemic: the emotional impacts
Coordinated by Michelle Kelly-Irving from the Epidemiology and Public Health Analysis: Risks, Chronic Diseases and Disability laboratory (Inserm/Université Toulouse III Paul-Sabatier), the Epidemic survey looks at the social and psychosocial factors of the epidemic and lockdown. It also aims to measure the psychological impact and the emotional, social and behavioral repercussions of the epidemic in order to prevent their consequences and develop appropriate support tools.
Lockdown, sleep, and dreams
The team of Perrine Ruby at the Lyon Neuroscience Research Center (Inserm/CNRS/Université Lyon 1/Université Jean Monnet Saint-Étienne) wishes to evaluate the impact of lockdown on our lifestyles, sleep, and dreams. The aim is to understand the impact on dream content and frequency during this particular period. The questionnaire is still accessible for all who wish to participate.
COVID Ethics
Led by Léo Coutellec from the Research in Ethics and Epistemology team at the Center for Research in Epidemiology and Population Health (Inserm/Université Paris-Saclay), this project involves analytical and critical commitment by the researchers to the COVID-19 epidemic in order to better understand and reinforce our framework of analysis of the ethical challenges in the anticipation and management of pandemic situations. This study will be conducted over 18 months and several seminars have been scheduled.
7. Test the population and protect and support medical staff
Inserm provides use of its laboratories
Inserm is participating in the population testing effort. A Decree and an Order published on April 5 authorize French prefects to requisition public research laboratories for COVID-19 testing using RT-PCR – a technique based on genetic material. For the laboratories under their supervision, the CNRS and Inserm have prepared for this by making inventories of the equipment available and the number of such tests that could be performed each day under the conditions set by the government.
Inserm launches a project to recycle masks
In close collaboration with Tours Regional University Hospital, Inserm Research Directors Nathalie Heuzé-Vourc’h and Mustapha Si-Tahar are leading a proof-of-concept study on an effective decontamination process for surgical and FFP2 masks, to enable their reuse. In the current context in which masks can be in short supply, various decontamination processes are being compared. The team has already shown that when 70°C moist heat is applied for a period of one hour, there is no deterioration in the structure of the masks. The decontamination appears to be effective, destroying several viruses and bacteria tested in the proof-of-concept study. Also, the properties of the masks are similar to the untreated masks. These findings must now be consolidated by testing the process on masks contaminated with SARS-CoV-2.
A project to develop an antiviral coating for people and surfaces
Inserm teams from the Biomaterials and Bioengineering unit (Inserm/Université de Strasbourg) and the Institute for Research on Viral and Hepatic Diseases (Inserm/Université de Strasbourg) have joined forces to work on antiviral coatings. This project involves developing an antiviral protection system that could be applied to various surfaces of medical devices and to people notably in the hospital environment – and which would be capable of destroying SARS-CoV-2 and limiting the transmission of the epidemic. The researchers intend to screen a number of molecules for their capacity to inhibit the virus, selecting those that are the most effective and with the most beneficial antiviral properties. The coatings will then be developed based on these molecules and their efficacy will be tested.
Aphro-Cov: strengthen the diagnosis and management of COVID-19 patients in five Sub-Saharan African countries
As part of the support given by France in response to the coronavirus crisis, Rémy Rioux, Chief Executive of the French Development Agency (AFD) and Gilles Bloch, Chairman and Chief Executive Officer of Inserm announced in March the launch of a joint initiative to improve the health surveillance and management of suspected cases of COVID-19 in five African countries (Burkina Faso, Gabon, Ivory Coast, Mali, Senegal). Called Aphro-Cov, this program steered by REACTing is focused on the laboratories, early warning system, clinical departments, and – by means of raised awareness and reinforced communication – the population as a whole.
Because research is needed more than ever to protect the health of everyone and to address the challenges posed by this new pandemic, Inserm has set up a platform for donations and is calling on public generosity. The funds collected will be used to sustain and complete these different research projects.
To make a donation and support the work of COVID-19 researchers: https://don.inserm.fr/donner
Inserm in the fight against fake news
Fighting false information and publishing clear and high-quality scientific information are key concerns for Inserm. This is particularly the case with the COVID-19 pandemic, given the various rumors and scientifically unfounded information circulating on social media and other platforms. To counteract this and give the public the most accurate information, Inserm has, since January 2020:
- Posted an episode on its Canal détox channel discussing the false information most often encountered.
- Published information bulletins with the most important news on the Institute and international research, and which also take a look at false information spotted online.
- Each week, the REACTing consortium prepares a thorough review of the scientific literature concerning SARS-CoV-2 and COVID-19. The group provides the public with a summary of high-quality scientific publications for those wishing to follow the scientific research more closely.

