During REM sleep, the brain inhibits the motor system, which makes the sleeper completely immobile. CNRS researchers working in the Centre de Recherche en Neurosciences de Lyon (CNRS/Université Claude Bernard Lyon 1/INSERM/Université Jean Monnet) have identified a population of neurons that is responsible for this transient muscle paralysis. The animal model created will shed light on the origin of some paradoxical sleep disorders, and more particularly the condition that prevents this paralysis. It will also be most useful in the study of Parkinson’s disease, since these pathologies are related. This work was published on December 12, 2016 on the website of the journal Brain.
In spite of being in a deep sleep, the patients talk, move, kick and eventually fall out of bed. They are suffering from a parasomnia called REM Sleep Behavior Disorder[1] (RBD). This disorder usually appears around the age of 50. Muscles are at rest during the REM sleep phase, but in these patients, there is no paralysis, although the reason for this is not known. The sleepers move abnormally, probably reflecting their dream activity.
A team from the Centre de Recherche en Neurosciences de Lyon (CNRS/INSERM/Université Claude Bernard Lyon 1/Université Jean Monnet) has taken one more step towards elucidating this pathology. The researchers identified neurons in the sublaterodorsal nucleus of the brain, ideally located to control motor system paralysis during REM sleep. In rats, they specifically targeted this neuron population, by adding genetically modified viral vectors to it.[2] Once these are in the neural cells, they block the expression of a gene that allows synaptic glutamate secretion. Now incapable of releasing this excitatory neurotransmitter, the neurons can no longer communicate with their neighbors. They are disconnected from the cerebral network necessary for paralysis during REM sleep.
For 50 years, the scientific community has considered that these glutamate neurons generated REM itself. This team’s experience invalidates this hypothesis: despite the absence of activity in this neuron circuit, the rats still experience this stage of sleep. They are fast asleep and disconnected from the outside world, with eyes closed. But these rats are no longer paralyzed. Their behavior is very reminiscent of the clinical profile of patients suffering from RBD. The glutamate neurons targeted in this study play an essential part in REM paralysis during sleep and are reportedly the first neurons affected in this neurological disease.
This research work goes beyond creating a new preclinical model that mimics this parasomnia. It may be of paramount importance in studying some neurodegenerative diseases. Recent clinical research has shown that patients diagnosed with RBD almost always develop the motor symptoms of Parkinson’s disease, on average a decade later. The team is now attempting to develop an animal model that evolves from parasomnia into Parkinson’s disease, in order to understand how neuron degeneration occurs.
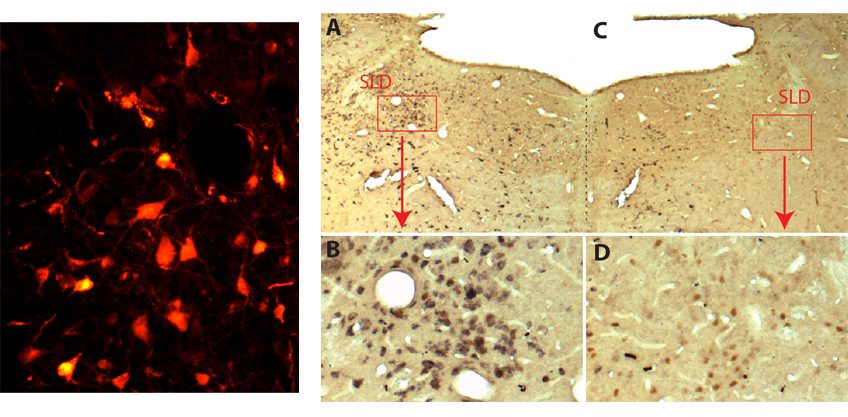
(Left) the glutamate neurons of the sublaterodorsal nucleus emit a spontaneous red fluorescence indicating that the viral vectors used have been successfully added. © Sara Valencia Garcia / Patrice Fort, CNRS
(right) in a normal rat specimen (A and B) the neurons of the sublaterodorsal nucleus (SLD, colored in brown) are glutamate neurons (also colored in black). In rats treated with viral vectors (C and D), neurons are still present (in brown) but are not longer capable of releasing glutamate (absence of black color). © Sara Valencia Garcia / Patrice Fort, CNRS
[1] REM sleep is also called paradoxical sleep.
[2] Provided by researchers at Tsukuba University in Japan