Press Contact
Inserm
Trinity College Dublin
Thomas Deane
Media Relations Officer
Public Affairs and Communications
Email: ei.dct@htenaed | Tel: 01 896 4685
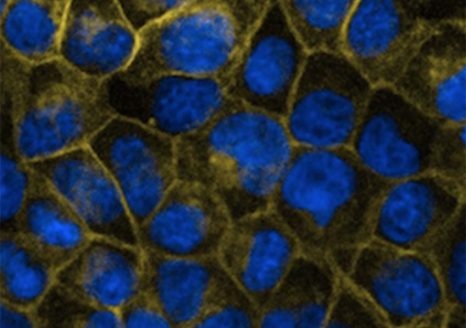
The effect of G60R mutation on CLDN-5 expression and localization in CLDN-null cells.
Scientists at Trinity College Dublin and the Institute Imagine at Necker Hospital, Paris, announced a significant advance in our understanding of a very rare condition called alternating hemiplegia of childhood (AHC). This is a devastating condition that can lead to repeated paralysis that affects one side of the body or the other or sometimes both at once. It usually begins to affect children before 18 months of age and to date, only one causative gene has been identified.
Here, scientists in Dublin and Paris have now identified a second gene CLDN5 as being responsible for the condition in 2 unrelated cases of AHC in France. The protein product of this gene, claudin-5, is critical for maintaining the integrity of the blood-brain barrier (BBB). Intriguingly, the mutated form of the protein turns the barrier into a channel that is selective for negatively charged ions. In this regard, the ionic compositions of the brain are likely shifted in these children and this is a key driver of the condition.
“This finding was based on an amazing collaboration with Prof Arnold Munnich’s group at the Institute Imagine in Paris. The identity of these de novo mutations in unrelated children suggests that the barrier is turning into a channel. This is exciting on numerous levels as it is the first report of the BBB turning into a channel, but it also sheds light on the devastating pathology of AHC which may assist in clinical management of patients with this mutation” said Dr Matthew Campbell, Associate Professor at Trinity.
Dr Yosuke Hashimoto, visiting researcher from the Japanese Society for the Promotion of Science (JSPS), and his equally contributing colleague Dr Karine Poirier from the Institute Imagine added: “This exciting project has shed light on a very rare condition affecting children. We are delighted that our work was able to quickly identify the causative mutation for the disease as well as progressing our understanding of the pathology of the disease.”
Commenting on the study, Prof Arnold Munnich from the Institute Imagine in Paris said “We are delighted this work has progressed so quickly and our groups have been able to work very closely to identify the cause of this condition. Studies like this will benefit families and clinicians immensely in the years to come”.
A multidisciplinary team of Geneticists, Neurologists and Radiologists from Ireland and France undertook the study.
Inserm
Trinity College Dublin
Thomas Deane
Media Relations Officer
Public Affairs and Communications
Email: ei.dct@htenaed | Tel: 01 896 4685
Recurrent de novo mutations in CLDN5 induce an anion-selective blood–brain barrier and alternating hemiplegia
Yosuke Hashimoto,1,† Karine Poirier,2,† Nathalie Boddaert,3 Laurence Hubert,2 Melodie Aubart,4 Anna Kaminska,4 Marianne Alison,5 Isabelle Desguerre,4 Arnold Munnich2,4,‡ and Matthew Campbell1,‡ †,
‡These authors contributed equally to this work.
1 Smurfit Institute of Genetics, Trinity College Dublin, Dublin 2, Ireland
2 INSERM UMR1163, Institut Imagine, Université Paris Cité, F-75015, Paris France
3 Department of pediatric radiology, Hospital Necker Enfants Malades, France
4 Departments of pediatric neurology and medical genetics, Hospital Necker-Enfants Malades, Université Paris Cité, F-75015, Paris France
5 Department of pediatric radiology, Hospital Robert Debré, Université Paris Cité, F-75015, Paris France
Brain https://doi.org/10.1093/brain/awac215 juin 2022