Why are we not aware of external noises while we sleep? A study carried out at NeuroSpin (French Atomic Energy Commission [CEA]/Inserm), in collaboration with the Sleep and Alertness Centre at Hôtel-Dieu Hospital, Paris (AP-HP), the Brain and Spinal Cord Institute (ICM), Collège de France, and Paris-Sud and Paris Descartes Universities, has shown that even though sounds continue to penetrate the auditory cortex, sleep disrupts the brain’s ability to anticipate them. The researchers have demonstrated that the brain is no longer capable of making predictions during sleep, because the predictive signals coming from the higher cortical areas seem to be eliminated. These results are published in the American journal PNAS, on 2 March 2015.
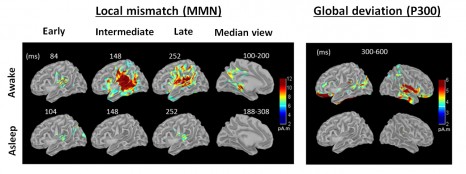
While listening to a melody during wakefulness, the brain uses the regularities in the sound sequence to predict the future sounds. This predictive ability is based on the functioning of a hierarchy of areas in the brain. If a sound breaks the regularity of the sequence, the brain then generates a series of “prediction error” signals responsible for, among other things, reactions to novelty or reactions of surprise. Previous studies using electroencephalography (EEG) have made it possible to describe at least two consecutive error signals, mismatch negativity (MMN) and P300. MMN has already been observed in subjects in an unconscious state (including a comatose state), whereas P300 may be specific for conscious processing, since it reflects the integration of information over a vast brain network, beyond the auditory regions.
During sleep, ambient sounds are not consciously perceived. However, we do not know the extent to which the integration of these sounds by the brain is disrupted, or whether the brain remains capable of perceiving their regularities and anticipating them. This particular aspect of brain function was tested by the NeuroSpin team (Inserm/CEA), in collaboration with the Sleep and Alertness Centre at Hôtel-Dieu Hospital, Paris (AP-HP), the Brain and Spinal Cord Institute (ICM), Collège de France, and Paris-Sud and Paris Descartes Universities. The researchers used electro- and magnetoencephalography (E/MEG) to study the prediction error signals (MMN and P300) in subjects during sleep and wakefulness.
The researchers invited volunteers to go to sleep inside NeuroSpin’s magnetoencephalography machine, in the presence of repetitive sounds. Results confirmed that P300 is a specific marker for conscious processing of sounds, since it disappeared as soon as the volunteers fell asleep, from which time the subjects no longer reacted to sounds. In contrast, MMN was observed at all stages of sleep (slow-wave sleep and rapid eye movement [REM] sleep). However, this signal is only partially retained, since some areas of the brain, which are normally activated during wakefulness, no longer respond to the sound stimulus. Indeed, the peak of activity resulting from a predictive error in an awake individual disappears during sleep. The only remaining phenomena are those of passive sensory adaptation, and confined to the primary auditory areas.
The researchers have thus shown that, due to a defect in communication between its different areas, the brain can no longer make predictions during sleep.
However, it remains capable of representing sounds within the auditory areas, and can get used to them if they are frequent, explaining why we are awakened by an alarm but not by the ticking of a clock.

©S. Dehaene. Reconstruction of the sources of error signals in the brain based on magnetoencephalographic recordings. The signals that indicate predictive error; the intermediate component of MMN and P300, disappear during sleep. Only passive mechanisms for sensory adaptation (the early and late components of MMN), confined to the auditory areas, remain. (Times are expressed in milliseconds, and measure the time taken to respond to the sound).
These contents could be interesting :