Researcher Contact
Christian Lesterlin
Chercheur Inserm
Laboratoire de microbiologie moléculaire et biochimie structurale (MMSB)
CNRS-UMR5086-Lyon 1
Tel: 04 72 72 26 64
Site internet:
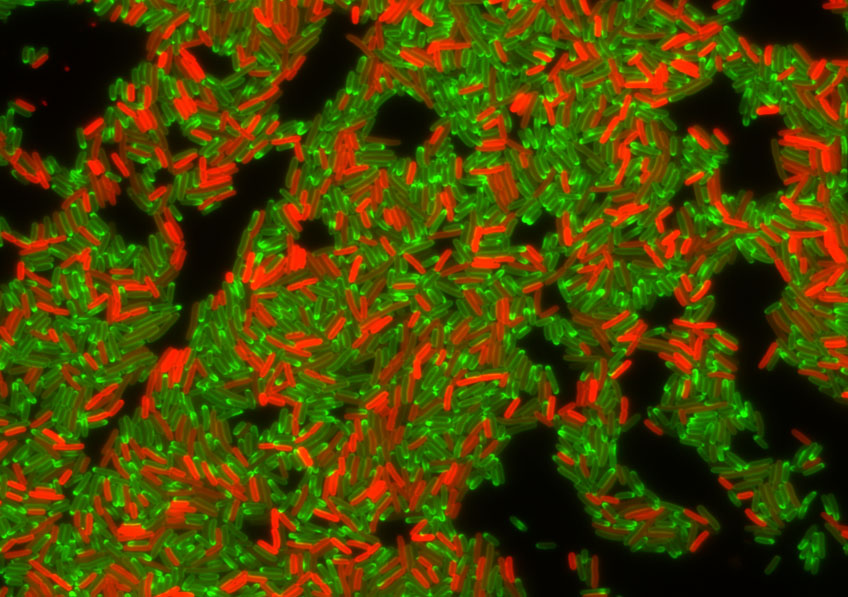
Drug resistance in bacterial population. Population of drug resistant bacterial cells producing a fluorescent labelled resistance factor, TetA efflux pump (in red), during treatment with tetracycline antibiotic (in green). Cells producing high levels of TetA efflux pump a capable or effective drug efflux and contain very little tetracycline drug. By contrast, cells containing a lot of tetracycline fail to produce TetA efflux pump.©Christian Lesterlin
The global spread of antibiotic resistance is a major public health issue and a priority for international microbiology research. In his paper to be published in the journal Science, Christian Lesterlin, Inserm researcher at Lyon’s “Molecular Microbiology and Structural Biochemistry” laboratory (CNRS/Université Claude Bernard Lyon 1), and his team were able to film the process of antibiotic resistance acquisition in real time, discovering a key but unexpected player in its maintenance and spread within bacterial populations.
This spread of antibiotic resistance is for the most part due to the capacity of bacteria to exchange genetic material through a process known as bacterial conjugation. The systematic sequencing of pathogenic or environmental strains has identified a wide variety of genetic elements that can be transmitted by conjugation and that carry resistance to most – if not all – classes of antibiotics currently used in the clinical setting. However, the process of transferring genetic material from one bacterium to another in vivo, the time needed to acquire this resistance once the new genetic material is received and the effect of antibiotic molecules on this resistance remained unelucidated.
Real-time visualization
The researchers chose to study the acquisition of Escherichia coli resistance to tetracycline, a commonly used antibiotic, by placing a bacterium that is sensitive to tetracycline in the presence of one that is resistant. Previous studies have shown that such resistance involves the ability of the bacterium to expel the antibiotic before it can exert its destructive effect using “efflux pumps” found on its membrane. These specific efflux pumps are able to eject the antimicrobial molecules from the bacteria, thereby conferring on them a certain level of resistance.
In this experiment, the transmission of the DNA from one specific “efflux pump” – the TetA pump – was observed between a resistant bacterium and a sensitive bacterium using fluorescent marking. Thanks to live-cell microscopy, the researchers just had to track the progression of the fluorescence to see how the DNA of the “pump” migrated from one bacterium to another and how it was expressed in the recipient bacterium.
The transfer of DNA from the donor bacteria (green) to the recipient bacteria (red) is revealed by the appearance of red localization foci. The rapid expression of the newly acquired genes is revealed by the production of green fluorescence in the recipient bacteria.
How is resistance organized in the presence of an antibiotic?
Tetracycline’s mode of action is well-known to scientists: it kills bacteria by binding to their translational machinery, thereby blocking any possibility of producing proteins. Following this line of reasoning, it would be expected that by adding the antibiotic to the previous culture medium, the TetA efflux pump would not be produced and the bacteria would die. However, the researchers observed that, paradoxically, the bacteria were able to survive and efficiently develop resistance, suggesting the implication of another factor essential to the process of acquiring resistance.
The scientists discovered that this phenomenon can be explained by the existence of another efflux pump that is present in virtually all bacteria: AcrAB-TolC. While this generalist pump is less efficient than TetA, it is still able to expel a small amount of antibiotic from the cell, meaning that the bacteria can maintain minimal protein synthesis activity. Therefore, if the bacterium is lucky enough to have received a resistance gene through conjugation, then the TetA pump is produced and the bacteria becomes durably resistant.
This study opens up new avenues in the search for similar mechanisms in bacteria other than E. coli, and for different antibiotics. “We could even consider a therapy combining an antibiotic and a molecule able to inhibit this generalist pump. While it is still too soon to envisage the therapeutic application of such an inhibitor, numerous studies are currently being performed in this area given the possibility of reducing antibiotic resistance and preventing its spread to the various bacterial species” concludes Lesterlin.
Christian Lesterlin
Chercheur Inserm
Laboratoire de microbiologie moléculaire et biochimie structurale (MMSB)
CNRS-UMR5086-Lyon 1
Tel: 04 72 72 26 64
Site internet:
Protecting role of AcrAB-TolC multidrug efflux pump in drug- resistance acquisition by plasmid transfer
Sophie Nolivos, Julien Cayron, Annick Dedieu, Adeline Page, Frederic Delolme, Christian Lesterlin.
Molecular Microbiology and Structurale Biochemistry (MMSB), Université Lyon 1, CNRS, INSERM, UMR5086, 69007, Lyon, France
Protein Science Facility, SFR BioSciences CNRS UMS3444, Inserm US8, UCBL, ENS de Lyon, 50 Avenue Tony Garnier, 69007 Lyon, France.
Science, https://dx.doi.org/10.1126/science.aav6390 24 mai 2019