©AdobeStock
In humans, “Theory of Mind” is the ability to understand others’ mental states: what they think, what they feel, what they desire, what they love, etc. It plays a major role in human social interactions.
But where did this ability evolve from? What type of selection pressure ultimately led to this being imparted on the human species?
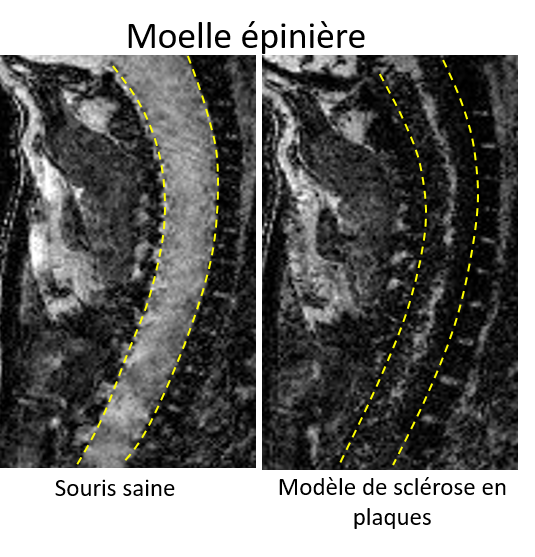
In order to offer a response, Jean Daunizeau, Inserm Researcher within ICM, Shelly Masi (National Museum of Natural History, MNHN) and her colleagues first developed a means of measuring the level of sophistication of theory of mind, based on behavioral analysis in simple interactive games. After validating this method in humans, they then used it to compare the level of sophistication of theory of mind in seven non-human primate species, from lemurs to the great apes (gorillas, orangutans, and chimpanzees).
Their study is the first to provide data on the origins of human social intelligence. In particular, the results of the study go against the generally accepted hypothesis, which stipulates that theory of mind developed in response to problems arising from the complexity of the social group in which animals evolved.
It appears rather that the evolution of theory of mind was primarily determined by limiting neurobiological factors, such as brain size.
Lastly, the researchers identified a large difference, an evolutionary “gap“, between the capacity of theory of mind in the great apes and humans. This research has been published in PLOS Computational Biology