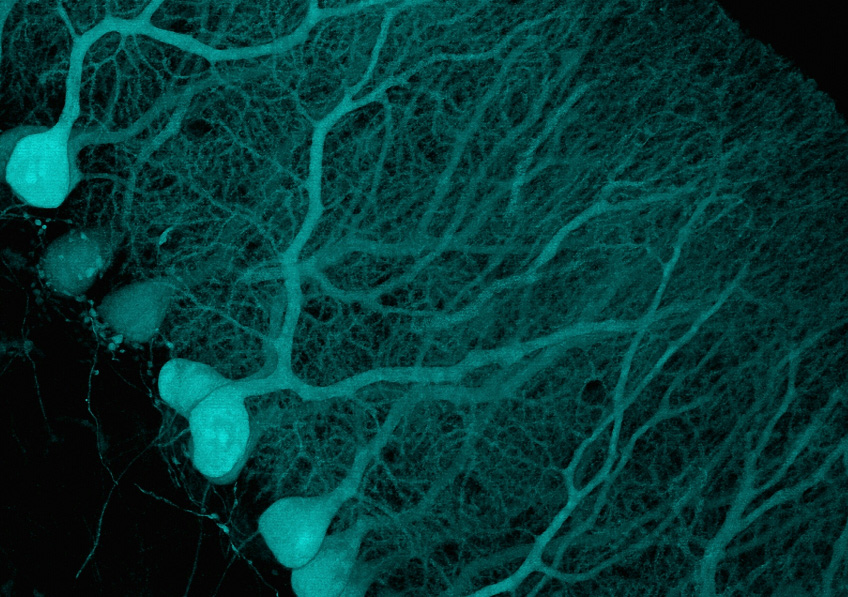
Restoring brain cholesterol turnover improves autophagy and has therapeutic potential in mouse models of spinocerebellar ataxias
Clévio Nóbrega1,2,3,4, Liliana Mendonça4, Adriana Marcelo1,2 , Antonin Lamazière5, Sandra Tomé4 , Gaetan Despres5 , Carlos A. Matos1,2,4 , Fatich Mechmet1,2, Dominique Langui6, Wilfred den Dunnen7, Luis Pereira de Almeida4,10, Nathalie Cartier8,9 , Sandro Alves11
1 Department of Biomedical Sciences and Medicine, University of Algarve, Faro, Portugal 2 Centre for Biomedical Research, University of Algarve, Faro, Portugal
3 Algarve Biomedical Center, University of Algarve, Faro, Portugal
4 Center for Neuroscience and Cell Biology, University of Coimbra, Coimbra, Portugal
5 INSERM, Saint-Antoine Research Center, Sorbonne Université, Faculté de Médecine, AP-HP, Hôpital Saint Antoine, Département PM2, Paris, France
6 Institut du Cerveau et de la Moelle épinière, ICM, INSERM U1127, CNRS UMR7225, Sorbonne Université, Hôpital Pitié-Salpêtrière, 47 bd de l’Hôpital, 75013 Paris, France
7 Department of Pathology and Medical Biology, University Medical Center Groningen, University of Groningen, PO Box 30.001, 9700 RB Groningen, The Netherlands
8 INSERM U1169 92265 Fontenay aux Roses and Université Paris-Sud, Université Paris Saclay, 91400 Orsay, France
9 Present Address: INSERM U1127, Institut du Cerveau et de la Moelle épinière (ICM), Hôpital Pitié- Salpêtrière, 47 bd de l’hôpital, 75013 Paris, France
10 Faculty of Pharmacy, University of Coimbra, 3000-548 Coimbra, Portugal
11 Brainvectis, Institut du Cerveau et de la Moelle épinière (ICM), Hôpital Pitié-Salpêtrière, 47 boulevard de l’Hôpital Paris, 75646 Paris, CEDEX 13, France
Acta Neuropathologica : https://link.springer.com/article/10.1007%2Fs00401-019-02019-7