Researcher Contact
Nicolas Gaudenzio
Unité différenciation épithéliale et autoimmunité
(UDEAR – Inserm / UT3 Paul Sabatier)
Téléphone : +33 (0)5 62 74 45 04
E-mail : rf.mresni@oizneduag.salocin
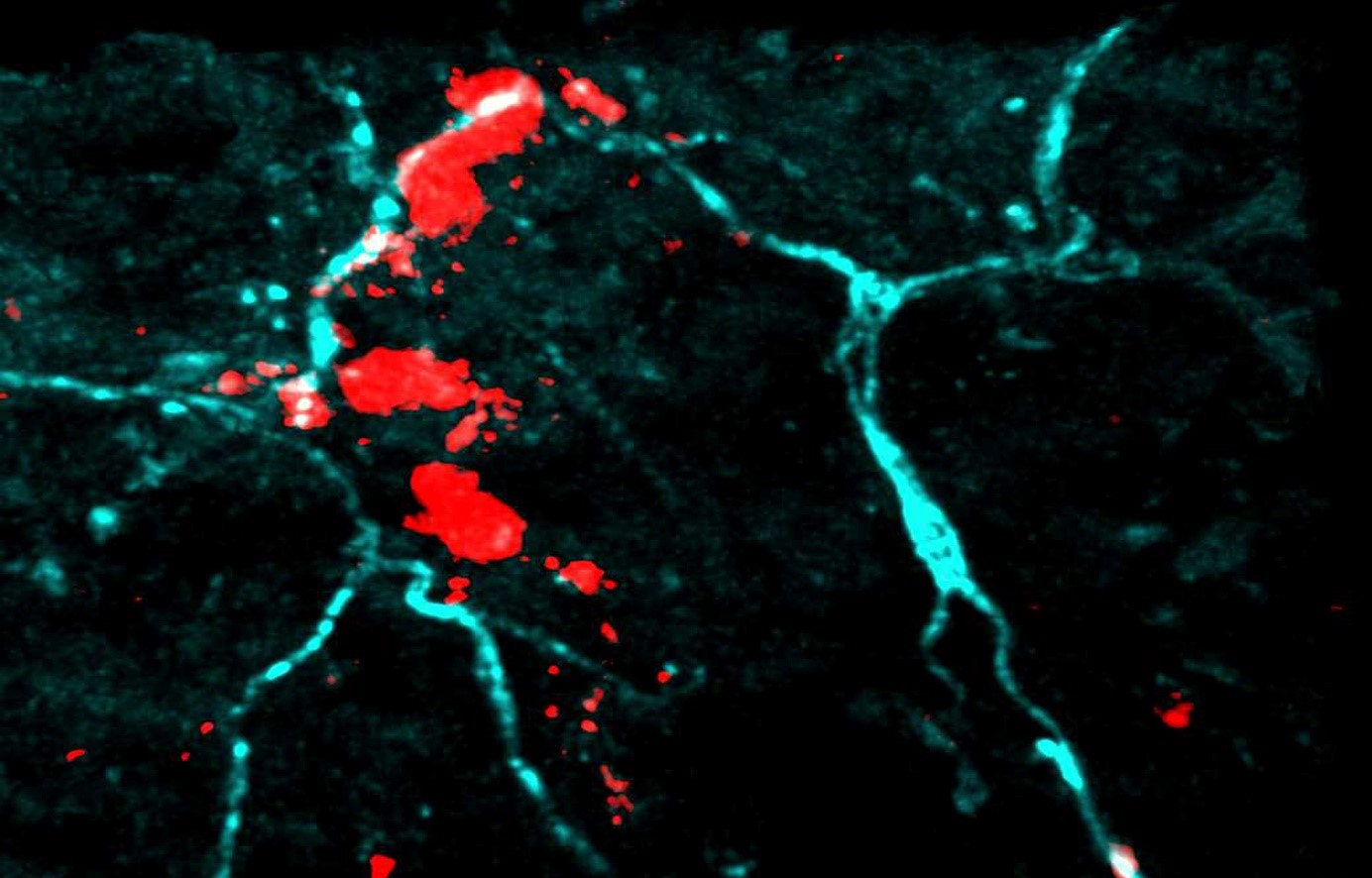
Mast cells and sensory neurons cluster in “bunches” under the skin. ©Nicolas Gaudenzio
Atopic dermatitis, or eczema, primarily affects infants and children, and manifests itself in hypersensitivity to allergens in the environment. A skin disease characterized by flare-ups, it is often treated with topical anti-inflammatories. A new study led by Inserm researcher Nicolas Gaudenzio, from the Epithelial Differentiation and Rheumatoid Autoimmunity Unit (UDEAR – Inserm / UT3 Paul Sabatier), in collaboration with his colleagues at Stanford University (United States) shows that immune cells and sensory neurons interact in the skin to form units that can detect allergens and trigger inflammation. A discovery that provides an insight into how atopic dermatitis works, and points the way to new therapeutic possibilities. Their findings have now been published in the journal Nature Immunology.
Dry skin, pain, and itching… Atopic dermatitis affects the everyday lives of nearly 20% of children, and up to 5% of adults. The condition can have a significant impact on the quality of life of these patients.
Several studies have shown that genetic factors are involved in the development of this chronic inflammatory skin disease, and suggest that they result in impairment of the skin barrier. This enables the allergens present in the environment, from pollen to dust mites, to penetrate the dermis and stimulate the immune system, which reacts abnormally to this “threat” by triggering eczema.
In particular, the researchers have taken a lead from existing clinical data which show that patients with this disease have numerous neuropeptides in their blood: chemical messengers that carry nerve messages, and whose level is correlated to disease severity. Identification of these neuropeptides in the blood indicates activation of the sensory neurons. These patients also have a number of enzymes in the blood indicating the presence of mast cells. Mast cells are immune cells present in the skin that play an essential role in modulating inflammatory and allergic processes.
Based on these observations, Gaudenzio and his team decided to focus on the interaction between sensory neurons and mast cells, and have now published their findings in the scientific journal Nature Immunology.
The researchers then showed that when the mice were exposed to dust mites, these “sensory neuroimmune units” were able to detect the presence of these allergens, triggering allergic inflammation.
In the longer term, this discovery could have practical therapeutic implications. “Until now, patients could be treated with biological treatments (biological therapy), but these obviously treat the disease further down the line, after flare-ups have occurred. We believe we have put our finger on a trigger mechanism and now want to continue our research to identify new molecules that could block interactions between mast cells and sensory neurons, and thus have a beneficial therapeutic effect for patients,” explains the researcher.
To do so, the group will first need to characterize the molecular interactions within these units in more detail, and analyze the role they play in modulating the immune system.
“One of the questions we are now going to try and answer is what these mast cell-sensory neuron units are for. They must represent a defense mechanism for the body, since they are also found in healthy individuals. But it could be that they don’t work properly in people who have atopic dermatitis—that’s what we’re trying to understand,” concludes Gaudenzio.
Nicolas Gaudenzio
Unité différenciation épithéliale et autoimmunité
(UDEAR – Inserm / UT3 Paul Sabatier)
Téléphone : +33 (0)5 62 74 45 04
E-mail : rf.mresni@oizneduag.salocin
House dust mites activate nociceptor–mast cell clusters to drive type 2 skin inflammation
Nadine Serhan 1,*, Lilian Basso 1,*, Riccardo Sibilano2,3, Camille Petitfils4, James Meixiong 5, Chrystelle Bonnart4, Laurent L. Reber6,7,8, Thomas Marichal8,9, Philipp Starkl10,11, Nicolas Cenac4, Xinzhong Dong 5,12, Mindy Tsai2,3, Stephen J. Galli 2,3,13,14 and Nicolas Gaudenzio 1,14
1 Unité de Différenciation Epithéliale et Autoimmunité Rhumatoïde, UMR 1056, INSERM, Université de Toulouse, Toulouse, France.
2 Department of Pathology, Stanford University School of Medicine, Stanford, CA, USA.
3 Sean N. Parker Center for Allergy and Asthma Research, Stanford University School of Medicine, Stanford, CA, USA.
4 IRSD, INSERM, INRA, INP-ENVT, Université de Toulouse, France
5 The Solomon H. Snyder Department of Neuroscience, Center for Sensory Biology, Johns Hopkins University School of Medicine, Baltimore, MD, USA.
6 Department of Immunology, Unit of Antibodies in Therapy and Pathology, Institut Pasteur, INSERM, U1222, Paris, France.
7 Center for Pathophysiology Toulouse Purpan, INSERM U1043, CNRS UMR 5282, Toulouse III University, Toulouse, France.
8 GIGA Institute and Faculty of Veterinary Medicine, Liege University, Liege, Belgium.
9 WELBIO, Walloon Excellence in Life Sciences and Biotechnology, Wallonia, Belgium.
10 CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences, Vienna, Austria.
11 Laboratory of Infection Biology, Department of Medicine I, Medical University of Vienna, Vienna, Austria.
12 Howard Hughes Medical Institute, Johns Hopkins University School of Medicine, Baltimore, MD, USA.
13 Department of Microbiology & Immunology, Stanford University School of Medicine, Stanford, CA, USA.
14, Corresponding authors: Stephen J. Galli, Nicolas Gaudenzio.
* These authors contributed equally: Nadine Serhan, Lilian Basso
Nature Immunology. Novembre 2019