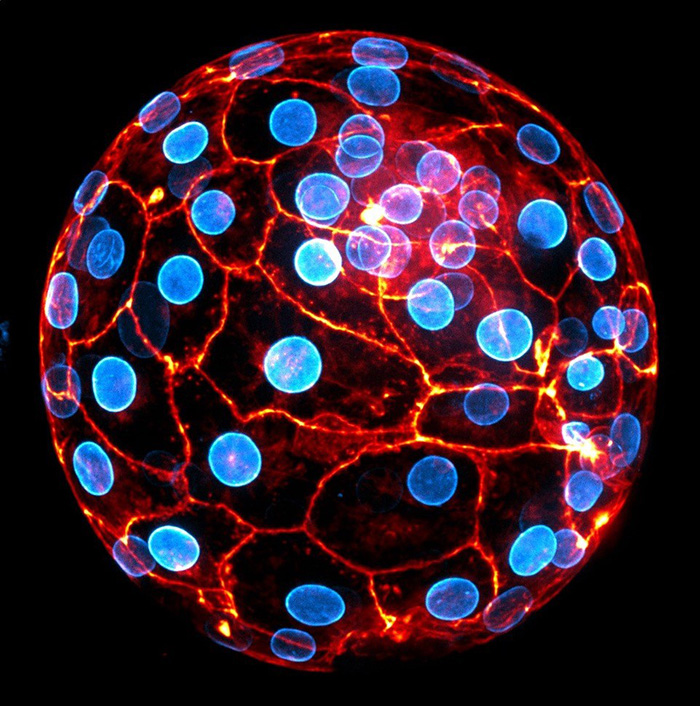
 Human embryo at the blastocyst stage ready to implant. The nuclear envelope of the cells appears in blue and the actin cytoskeleton in orange. © Julie Firmin et Jean-Léon Maître (Institut Curie, Université PSL, CNRS UMR3215, INSERM U934)
Human embryo at the blastocyst stage ready to implant. The nuclear envelope of the cells appears in blue and the actin cytoskeleton in orange. © Julie Firmin et Jean-Léon Maître (Institut Curie, Université PSL, CNRS UMR3215, INSERM U934)
Human embryo compaction, an essential step in the first days of an embryo’s development, is driven by the contractility of its cells. This is the finding of a team of scientists from CNRS, Institut Curie, Inserm, AP-HP and the Collège de France. Published in the 1st May edition of Nature, these results contradict the presupposed driving role of cell adhesion in this phenomenon and pave the way for improved assisted reproductive technology (ART).
In humans, embryonic cell compaction is a crucial step in the normal development of an embryo. Four days after fertilisation, cells move closer together to give the embryo its initial shape. Defective compaction prevents the formation of the structure that ensures the embryo can implant in the uterus. In assisted reproductive technology (ART), this stage is carefully monitored before an embryo is implanted.
An interdisciplinary research team1 led by scientists at the Genetics and Developmental Biology Unit at the Institut Curie (CNRS/Inserm/Institut Curie) studying the mechanisms at play in this still little-known phenomenon has made a surprising discovery: human embryo compaction is driven by the contraction of embryonic cells. Compaction problems are therefore due to faulty contractility in these cells, and not a lack of adhesion between them, as was previously assumed. This mechanism had already been identified in flies, zebrafish and mice, but is a first in humans.
By improving our understanding of the early stages of human embryonic development, the research team hopes to contribute to the refinement of ART as nearly one third of inseminations are unsuccessful today.2
The results were obtained by mapping cell surface tensions in human embryonic cells. The scientists also tested the effects of inhibiting contractility and cell adhesion and analysed the mechanical signature of embryonic cells with defective contractility.

Human embryo at the 4-cell stage. Cell DNA appears in red and their actin cytoskeleton in blue. The cell on the right has just split its genome into two and is about to divide. © Julie Firmin et Jean-Léon Maître (Institut Curie, Université PSL, CNRS UMR3215, INSERM U934)
1 – Scientists from the following entities also took part in the study: the Centre interdisciplinaire de recherche en biologie (CNRS/Collège de France/Inserm), the Reproductive Biology Department – CECOS (AP-HP) and the Institut Cochin (CNRS/Inserm/Université Paris Cité).
2 – Source: Agence de la biomédecine
These contents could be interesting :
Mechanics of human embryo compaction.
Julie Firmin, Nicolas Ecker, Diane Rivet Danon, Özge Özgüç, Virginie Barraud Lange, Hervé Turlier, Catherine Patrat et Jean-Léon Maître.
Nature, 1st May 2024.

