Scientists from the Institut Pasteur, INSERM, Collège de France, and Pierre and Marie Curie University, in collaboration with a team from the University of Auvergne, identified mice models that mimic high-frequency hearing impairment in humans, with a strong low-frequency sound interference. Their work sheds light on the anomalies causing the hearing impairment and reveals cochlear defects that profoundly affect the way sound frequencies are processed. This work could explain the pronounced masking effect experienced by some hearing-impaired individuals when trying to discriminate high-frequency sounds in noisy environments. The scientists suggest that more substantial auditory assessments would enable clinicians to improve diagnosis of these auditory impairments and provide better care for individuals who, despite showing only a mild hearing impairment using standard audiometric evaluations, should be fitted with hearing aids that appropriately target the defective sound frequencies and correct the hearing impairment.
Despite an audiometric evaluation revealing mild hearing impairment only, some patients experience severe difficulties in understanding speech because of profound sound interference in noisy environments. This represents a major handicap in their daily lives. By studying mice carrying a mutation that affects a subpopulation of auditory hair cells, scientists were able to observe a clinical profile similar to that of these patients. The project was led by Dr. Aziz El-Amraoui and Professor Christine Petit, head of the Genetics and Physiology of Hearing Unit (Institut Pasteur/INSERM UMRS 1120/Pierre and Marie Curie University/Collège de France), in tight collaboration with the neurosensory biophysics team from the INSERM joint research unit UMR 1107 at the University of Auvergne led by Professor Paul Avan.
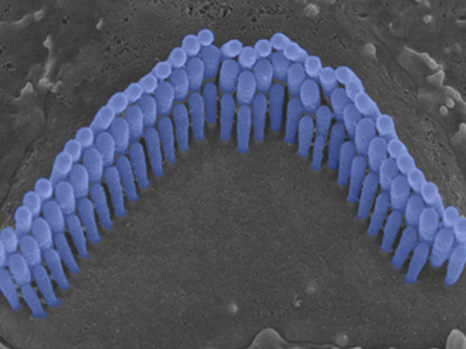
The mice being studied presented a misleadingly mild hearing loss, observed only for high-frequency sounds, sound frequencies that are normally processed at the cochlear base. However, scientists found significant morphological defects in this very area. The outer hair cells at the cochlear base were unable to respond to high-frequency sounds and their corresponding hair bundles, which act as antennas for sound reception, were markedly misshaped (see photo).
Subjecting the mice to interfering sounds led the scientists to discover an unexpected phenomenon: the low frequency sounds (two octaves lower, and at much lower intensity) significantly masked the high-frequency sounds. The scientists were then able to identify that the fully functioning cochlear apex, which normally processes low-frequency sounds, was behind the masking effect. However, in the mutant mice where the cochlear base had lost all sensitivity, the cochlear apex encoded both low- and high-frequency sounds. In a healthy cochlea, high-frequency sounds cannot even reach the apical region because of the base-to-apex varying physical characteristics of the basilar membrane. However, hair bundle anomalies in mutant mice seem to allow high-frequency sound vibrations to travel along another pathway with very different physical characteristics.
Transposed to humans, this study suggests that some individuals with seemingly optimistic audiograms should indeed be urged to use hearing aids. To avoid the misleading interpretations of standard audiometric tests, individuals with high–frequency hearing impairment vulnerable to interference by low-frequency sounds should undergo complementary clinical evaluations. This would notably allow for a more detailed study of the frequency responses of their auditory sensory cells. Accordingly, the prescribed auditory hearing aids should appropriately focus on the selective restoration of high-frequency sound detection, while taking into account the prevention of low-frequency sound interference.
Illustration: Hair bundles of the auditory outer hair cells from a normal mouse (upper) and a mutant mouse (lower) observed by scanning electron microscopy. – Copyright Institut Pasteur – V. Michel & K. Kamiya
This study was funded by the ERC-hair bundle grant (2011-ADG_20110310), LABEX Lifesenses, the Japan Society for the Promotion of Science, the Uehara Memorial Foundation, Réunica-Prévoyance, Humanis, Irène Errera-Hoechstetter, the Fondation BNP-Paribas and the Fondation Voir et Entendre.
These contents could be interesting :
Proceedings of the National Academy of Sciences of the United States of America (9 juin 2014).Kazusaku Kamiya (a,b,c,d,1), Vincent Michel (a,b,c,1), Fabrice Giraudet (e), Brigitte Riederer (f), Isabelle Foucher (a,b,c), Samantha Papal (a,b,c), Isabelle Perfettini (a,b,c), Sebastien Le Gal (a,b,c), Elisabeth Verpy (a,b,c), Weiliang Xia (f), Ursula Seidler (f), Maria-Magdalena Georgescu (g),Paul Avan (e,2,3), Aziz El-Amraoui (a,b,c,2,3), and Christine Petit (a,b,c,h,2,3)(a) Institut Pasteur, Génétique et Physiologie de l’Audition, 75015 Paris, France(b) Institut National de la Santé et de la Recherche Médicale, UMR-S 1120 Paris, France(c) Sorbonne Universités, UPMC Univ Paris 06, Complexité Du Vivant, 75252 Paris Cedex 05, France(d) Department of Otorhinolaryngology, Juntendo University Faculty of Medicine, Juntendo University, Tokyo 1138421, Japan(e) Laboratoire de Biophysique Sensorielle, INSERM UMR 1107, Faculté de Médecine, Université d'Auvergne; 63000 Clermont-Ferrand, France(f) Department of Gastroenterology, Hepatology, and Endocrinology, Hannover Medical School, 30625 Hannover, Germany(g) Department of Neuro-Oncology, The University of Texas MD Anderson Cancer Center, Houston, USACollège de France, 75005 Paris, France