On the occasion of International Rare Disease Day: 28 February 2013
How has research into Usher Syndrome – a particularly debilitating hereditary disease that damages the two main senses, hearing and sight – overcome a major hurdle in understanding the mechanisms causing pigmentary retinopathy? How has it managed to improve clinical diagnosis and develop molecular diagnosis? How has it prepared gene therapy tests for the retinopathy aspect of the syndrome? Scientists who have joined forces in the European TREATRUSH (TreatRetUsher) project have been reporting progress. Twelve partners in seven countries came together to form this network, financed by the European Commission and coordinated by the Pierre and Marie Curie University (UPMC). In France, it brings together researchers from the Collège de France, INSERM, the Institut Pasteur, the CNRS and the UPMC, working at the Institut de la Vision and the Institut Pasteur, as well as clinicians from the XV-XX Hospital and the Armand-Trousseau Hospital.
Usher Syndrome is a hereditary disease attacking hearing and sight. It affects about one person in 10,000. It is the main cause of deafness associated with a loss of vision in young people. The three clinical types of the syndrome, USH1, USH2 and USH3, are distinguished by the severity of hearing loss, early onset of retinitis pigmentosa and the presence or otherwise of a vestibular (inner ear) attack that translates into difficulties with balance. In each of these aspects of the disease the USH1 form is the most incapacitating.
Going beyond the major advances previously achieved in identifying the genes responsible for the syndrome and understanding the pathogenesis of the damage done to hearing by the syndrome, the Treatrush project has set itself the aim of improving diagnosis, understanding the cellular and molecular mechanisms behind retinopathy and preparing gene therapy trials.
- Responding to the imperative of early diagnosis.
A reduction in vision only manifests years after the loss of hearing, so the absence of early diagnosis of the syndrome can cause parents to make the wrong choice, and cause their children to learn sign language as young as possible, rejecting early cochlear implantation. Consequently, the European Consortium’s ENT specialists and ophthalmologists have jointly drawn up clinical investigation protocols that should result in a diagnosis being available for children under one year old. At the same time, a robust and swift molecular diagnosis of all forms of the syndrome has been developed. An initial study[1] enabled the detection of mutations in 54 patients. It was supplemented by the introduction of a new method[2] that now makes it possible to analyse a cohort of more than 350 patients recruited throughout Europe (France, Germany, Italy, Slovenia, Spain).
- Clarifying the physio-pathological mechanisms that cause retinopathy.
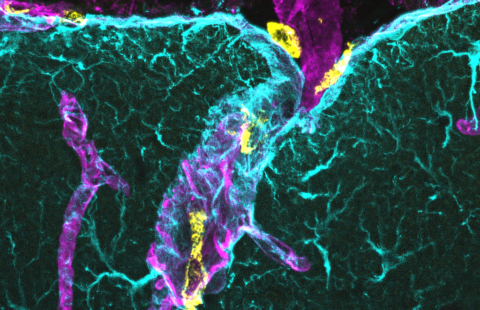
While mutant mice in which there are defective Usher-1 genes are profoundly deaf, they do not present with retinopathy. Consequently, the defective mechanisms causing retinopathy in those suffering from the syndrome were totally unknown. Three years ago, teams headed by Professor Christine Petit, co-ordinator of the Treatrush Project, and Professor José-Alain Sahel, attempted to understand the physio-pathological mechanisms causing retinitis pigmentosa in patients suffering from Usher Syndrome, by using other animal models[3]. They were successful and thus laid the foundation for discovering a new type of mechanism that causes pigmentary retinopathy, one that also involves the structures of photoreceptors, an aspect hitherto neglected, the role of which is still unknown. This data is essential for the creation of any protocol designed to improve treatment.
Project teams in Italy, France and the United States are currently working to develop gene therapy via viral vectors associated with the adenovirus (AAV) capable of effectively transferring tiny therapeutic genes into retinal photoreceptors with the aim of preventing their degeneration. These vectors have already proven their worth in other forms of retinitis. They are currently being tested on animals to identify some of the Usher genes.
The advances achieved in curing this rare illness open the way to an understanding and treatment of commoner causes of deterioration in vision and hearing. As Christine Petit and José-Alain Sahel explain, “frequent disorders are of a more complex origin and are thus harder to study than rare monogenic diseases. Yet many of the contributing factors should belong to the same elementary mechanisms as those which, when defective, lead to a particular, rarer disease. As proof, take the example of the USH1 Syndrome. The molecules encoded by these genes lie at the heart of the auditory transduction machinery, machinery that converts a sound signal into an electric signal in the auditory sensory cells. It is self-evident that in cases of frequent damage to hearing, regardless of origin, this machinery is also the target of deficiency. A constellation of rare diseases should thus contribute to creating the model for the pathogenic processes occurring in a common disease”.
This syndrome is a hereditary sensory disability and the main cause of combined loss of hearing and blindness. It is responsible for 3% to 6% of infant deafness and about 50% of cases of severe deafness associated with adult blindness. Deafness is generally congenital, while the clinical manifestations of retinitis pigmentosa occur slightly later in life. This syndrome has been divided into three sub-types, USH1, USH2 and USH3; the USH1 form is the most serious. In children suffering from the USH1 form, damage to sight is frequently not discovered until around the age of eight or ten. This late diagnosis makes it very hard to provide the appropriate medical treatment.
Important scientific progress was made by geneticists and hearing physiologists about fifteen years ago, when they identified the ten genes responsible and the discovery of deficient cellular and molecular mechanisms explained the loss of hearing caused by the syndrome. Conversely, the pathogenesis of retinitis pigmentosa in the syndrome remained unknown. A major advance occurred recently in understanding the damage caused to the retina, as part of the European Treatrush Project, through research conducted by teams headed by Professors Christine Petit and José-Alain Sahel. The patient exploration protocols were improved and standardised to allow for early diagnosis. A new molecular diagnostic tool was developed and more than 400 patients from various European countries were tested. In 1995, Professor Christine Petit’s laboratory identified the first gene causing the USH1 Syndrome. This is the gene that encodes VIIa myosin and is responsible for most cases of USH1. It is precisely in this form of the syndrome that a gene therapy retinopathy trial will soon be conducted by Professor José-Alain Sahel.
The aim of TREATRUSH is to treat and combat blindness caused by the Usher Syndrome. https://www.treatrush.eu
The project began in February 2010 and lasted for four years, the total budget of about 6 million euros being funded by the European Union (PC7). The project involves twelve partners based in seven countries:
Université Pierre and Marie Curie (UPMC), Paris, France: https://www.upmc.fr/
Institut National de la Santé et de la Recherche Médicale (Inserm), Paris, France : www.inserm.fr/
Eberhard Karl University of Tübingen (EKUT), Tübingen, Germany: https://www.uni-tuebingen.de/en
Medical Research Council (MRC), Oxford, UK: https://www.mrc.ac.uk
Fondazione Telethon (FTELE.IGM), Naples, Italy: https://www.telethon.it/
Amsterdam Molecular Therapeutics (AMT), Amsterdam, Netherlands: https://www.amtbiopharma.com/
Novartis Forschungsstiftung, Zweigniederlassung Friedrich Miescher-Institut for Biomedical Research, Basle, Switzerland: https://www.fmi.ch/
Faun Foundation (FAUN), Nuremberg, Germany
The Trustees of the University of Pennsylvania (UPENN), Philadelphia, USA : https://www.upenn.edu/
Institut de la Vision-Fondation Voir et Entendre, Paris, France: https://www.institut-vision.org https://www.fondave.org/
Johannes Gutenberg University of Mainz, Mainz, Germany: https://www.uni-mainz.de/eng/
Massachusetts Eye and Ear Infirmary, Bos
[1] Bonnet C, Grati M, Marlin S, Levilliers J, Hardelin JP, Parodi M, Niasme-Grare M, Zelenika D, Délépine M, Feldmann D, Jonard L, El-Amraoui A, Weil D, Delobel B, Vincent C, Dollfus H, Eliot MM, David A, Calais C, Vigneron J, Montaut-Verient B, Bonneau D, Dubin J, Thauvin C, Duvillard A, Francannet C, Mom T, Lacombe D, Duriez F, Drouin-Garraud V, Thuillier-Obstoy MF, Sigaudy S, Frances AM, Collignon P, Challe G, Couderc R, Lathrop M, Sahel JA, Weissenbach J, Petit C, Denoyelle F. (2011) Complete exon sequencing of all known Usher syndrome genes greatly improves molecular diagnosis. Orphanet J Rare Dis. 6:21.
[2] Fakin A, Jarc-Vidmar M, Glavač D, Bonnet C, Petit C, Hawlina M. (2012) Fundus autofluorescence and optical coherence tomography in relation to visual function in Usher syndrome type 1 and 2. Vision Res. 75: 60-70.
[3] Sahly I, Dufour E, Schietroma C, Michel V, Bahloul A, Perfettini I, Pepermans E, Estivalet A, Carette D, Aghaie A, Ebermann I, Lelli A, Iribarne M, Hardelin JP, Weil D, Sahel JA, El-Amraoui A, Petit C. (2012) Localization of Usher 1 proteins to the photoreceptor calyceal processes, which are absent from mice. J Cell Biol. 15;199(2):381-99.