Why does multiple sclerosis progress more rapidly in some patients than others? Why do some patients with MS manage to regenerate their myelin, while others do not? Inserm researchers at Unit 1127, “Brain and Spine Institute” (Inserm/CNRS/UPMC) have demonstrated that lymphocytes play a major role in the remyelination process, and that they could possibly be exploited to develop new myelin regeneration strategies.
This work is published in Brain
Multiple sclerosis (MS) is an inflammatory disease of the central nervous system, causing progressive destruction of the myelin sheath that surrounds the axons and is essential for their protection and for the transmission of nerve impulses. The ability to repair myelin efficiently is a key factor in preventing progression of the disease. It is essential to understand why and how certain patients manage to better handle the disease than others.
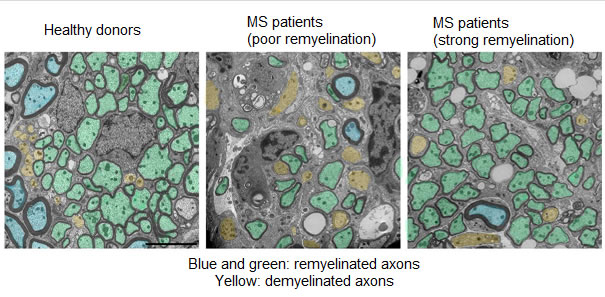
In multiple sclerosis, T lymphocytes attack myelin as if it were a virus, which is abnormal, but it is also they that organise its repair, more or less well. Lymphocytes activate macrophages and microglia (other cells of the immune system), which in turn attract new stem cells to the site of the lesion in order to repair the damaged myelin. Previous studies showed that in some patients, the lesions are completely repaired while in other patients, once the lesion has appeared, it is never repaired.
To gain a better understanding of the phenomenon, the researchers at Inserm transplanted lymphocytes from healthy donors or patients with multiple sclerosis into demyelinated lesions in the spinal cord of mice.
Using this technique, the researchers showed that the problem was not associated with the first phase of recruiting cells capable of repair, but with the point where these cells differentiate in order to perform myelin repair. In patients with a strong capacity for remyelination, the lymphocytes send the appropriate signals to activate the microglia, which then enter a state of activation and bring about stem cell differentiation and myelin repair. In patients with a poor capacity for remyelination, the T lymphocytes do not allow activation of the microglia, affecting the entire repair cascade.
By comparing the secretion profiles of lymphocytes from patients with a strong or poor capacity for remyelination, the researchers demonstrated the existence of 3 molecules associated with good remyelination, and 3 associated with poor remyelination.
One of these molecules is CCL19, which is associated with poor remyelination capacity. The researchers propose a hypothesis whereby inhibiting this molecule would allow macrophages to attain a state of activation and could therefore affect the remyelination profile of patients.
Since microglial cells and macrophages are essential elements in coordination and repair, these results could also contribute additional elements of information regarding other conditions such as amyotrophic lateral sclerosis (also known as motor neuron disease), Alzheimer’s disease and Parkinson’s disease.
“The study of lymphocytes from patients with strong capacities for remyelination is a promising route to the development of new myelin regeneration strategies. Moreover, the systematic study of their lymphocytes might make it possible to provide support for diagnosis and treatment and to develop precision medicine tailored to each patient,” explains Violette Zujovic, Inserm Researcher and main author of this work.