Press Contact
École Polytechnique
Sara Tricarico
+ 33 1 69 33 38 70
+ 33 6 66 53 56 10
ude.euqinhcetylop@ociracirt.aras
Raphaël de Rasilly
ude.euqinhcetylop@yllisar-ed.leahpar
+ 33 1 69 33 38 97
+ 33 6 69 14 51 56
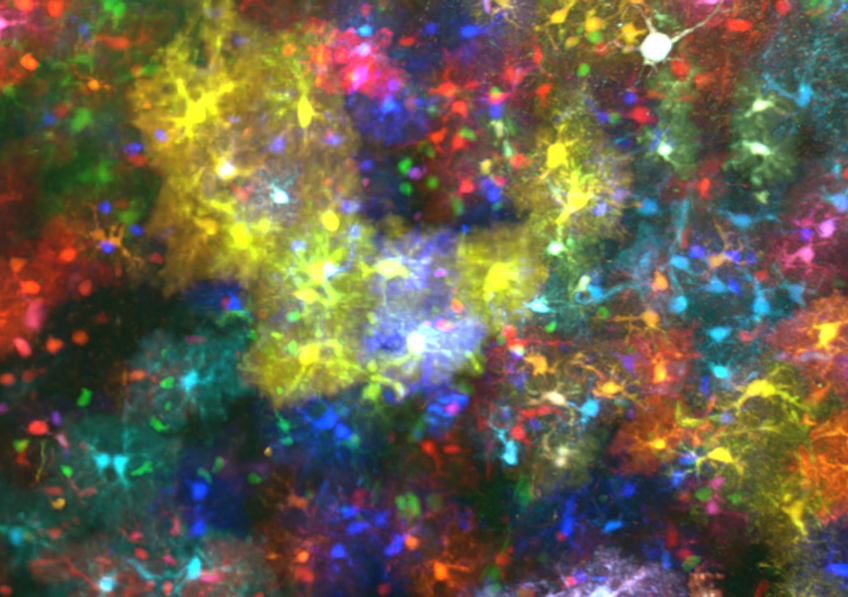
©3D brainbow astrocytes
Developed by researchers from École Polytechnique, Sorbonne Université, Inserm and the CNRS, ChroMS is a new microscopy technique bringing together color, 3D and high-resolution imaging, and is nothing short of a revolution in vertebrate brain imaging. The ChroMS technique is described in detail in a recently published article in Nature Communications.
Until now, researchers have had to choose between resolution and volume when performing vertebrate brain imaging. They could either obtain very high resolution over small volumes using three-dimensional electron microscopy or an image of the whole brain at resolutions that are far too low to understand the details.
“This instrument is ideal for making extremely precise, 3D reconstructions of regions of the brain with a volume of a few cubic millimeters, which is a breakthrough with this image quality, and this is the appropriate scale for what we want to observe”, explains Emmanuel Beaurepaire, researcher from the Laboratory for Optics and Biosciences (LOB – a joint research lab between École Polytechnique, the CNRS and Inserm). “Using the current version of our instrument, we can also reconstitute a complete mouse brain, albeit at a lower level of precision”.
“We are particularly interested in cell lineage,” states Jean Livet, researcher from the Institut de la Vision (Sorbonne Université, Inserm, CNRS). “In other words, the manner in which the brain develops from neural stem cells: what are the daughter cells from a given stem cell, how can a stem cell mutation influence their development, and how are groups of cells generated by different stem cells organized in relation to one another? The high-volume, color-coded images produced by ChroMS reveal the developmental history of an individual region of the brain”.
ChroMS should enable us to answer questions that neuroscientists have been asking for a long time, such as whether neurons arising from the same stem cell connect to each other preferentially to fulfil a given function, and whether pathologies such as epilepsy could be linked to localized problems affecting certain neural stem cells.
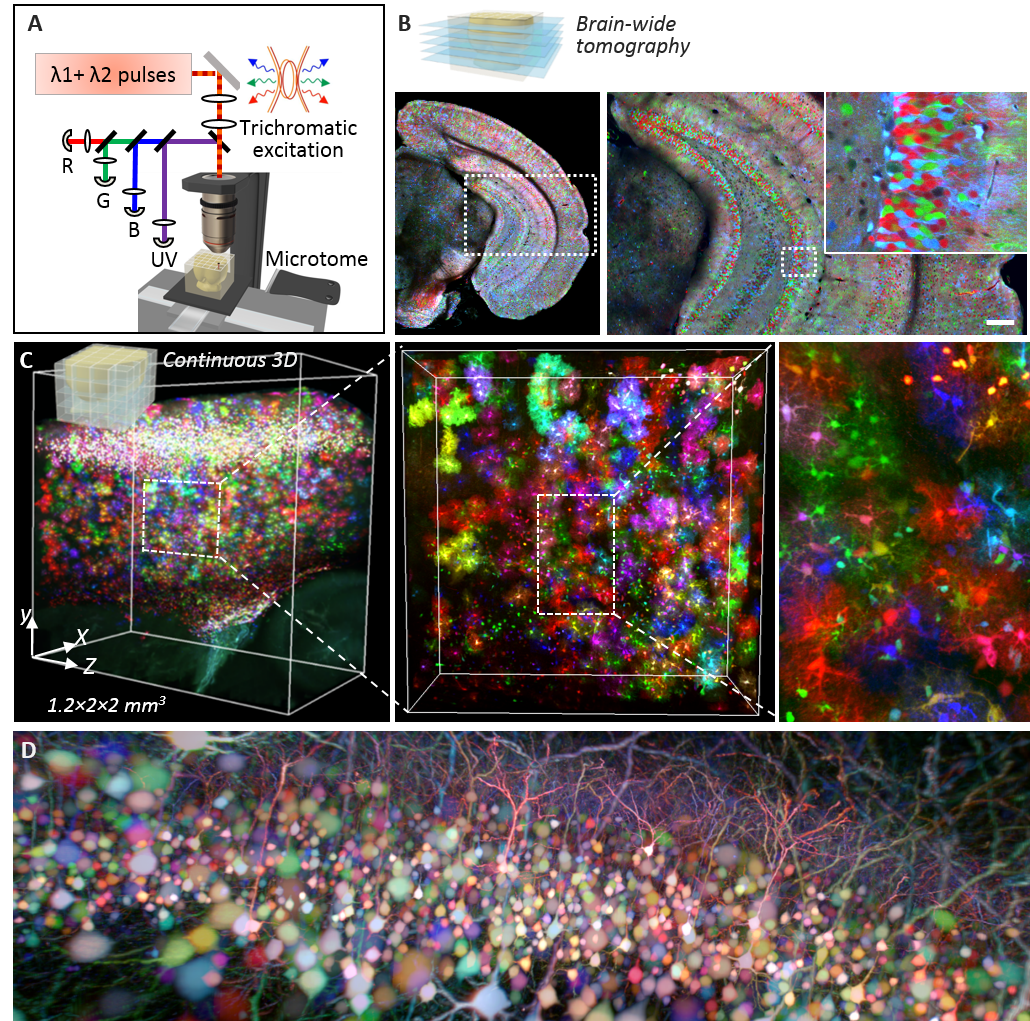
(A) Principle of ChroMS microscopy, combining color two-photon excitation by frequency mixing and automated serial slicing of brain tissue. (B) Image acquired with the “whole brain tomography” mode showing the cortex and hippocampus of a Brainbow mouse. (C) 3D reconstruction and view at different scales of a 4.8 mm3 volume of mouse cortex in which astrocytes are marked with fluorescent proteins of different colors. (D) 3D view of color-marked neurons in the mouse cortex. Adapted from: Abdeladim et al, Nat Commun 2019.
École Polytechnique
Sara Tricarico
+ 33 1 69 33 38 70
+ 33 6 66 53 56 10
ude.euqinhcetylop@ociracirt.aras
Raphaël de Rasilly
ude.euqinhcetylop@yllisar-ed.leahpar
+ 33 1 69 33 38 97
+ 33 6 69 14 51 56
“Multicolor multiscale brain imaging with chromatic multiphoton serial microscopy”
Lamiae Abdeladim, Katherine S. Matho, Solène Clavreul, Pierre Mahou, Jean-Marc Sintes, Xavier Solinas, Ignacio Arganda-Carreras, Stephen G. Turney, Jeff W. Lichtman, Anatole Chessel, Alexis-Pierre Bemelmans, Karine Loulier, Willy Supatto, Jean Livet, Emmanuel Beaurepaire. Nature Communications (2019).