© Fotolia
An international collaborative study coordinated by Frédéric Laumonnier (Unit 930 “Imaging and Brain” Inserm/University of Tours) and Yann Hérault of the Institute of Genetics and Molecular and Cellular Biology (Inserm/ CNRS/ University of Strasbourg) provides new and original findings on the pathophysiological role of the contact areas between neurons in certain brain disorders. The study reveals that mutation of one of the genes involved in intellectual disability and autism spectrum disorder leads to dysfunction of the synapses, which are essential for neuronal communication. The research was published on April 18, 2017, in Molecular Psychiatry.
Autism spectrum disorder (ASD) and intellectual disability (ID) are neurodevelopmental disorders that generally emerge when a child’s brain is developing and often persist into adulthood. Behavioral disorders and inabilities to communicate and establish social interactions are observed in people with ASD. In addition, those with ID present difficulties with comprehension, memory, and learning. While the origins of these disorders remain poorly understood, we now know that a significant proportion are associated with genetic mutations.
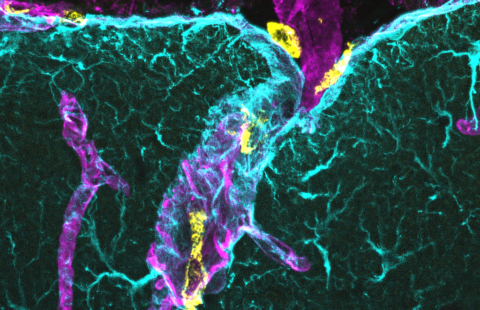
During the brain development process, synapse formation is essential for brain functions such as memory and learning. Synapses are the points of contact between neurons which enable neurons to connect with each other and propagate information. Some synapses are inhibitory and others excitatory, to enable the establishment of functional neuronal networks. However, mutations of the so-called PTCHD1 (Patched Domain containing 1) gene, which is located on the X chromosome and enables the expression of a protein potentially involved in synaptic functioning, have recently been identified in boys with the aforementioned disorders. These mutations stop the gene from expressing itself.
In order to validate the involvement of PTCHD1 gene mutations in ASD and ID, Hérault and his co-workers created a mouse model that was deficient for the PTCHD1 gene. In these animals, they observed major memory deficits and significant symptoms of hyperactivity, thus confirming the gene’s involvement in ASD and ID. Parallel studies by Laumonnier’s team showed a presence of the PTCHD1 protein in the excitatory synapses and also detected changes in the same mice’s synapses.
These changes to synaptic structure and activity in the excitatory neuronal networks were found to be particularly significant in a central brain region known as the hippocampus. This region plays a major role in cognitive processes, particularly those involving memory and the formation of new memories.
Genetic abnormalities impacting the structure or functioning of these synapses constitute a pathophysiological target in ASD and ID. In this context, this research defines a new “synaptic disease” caused by a PTCHD1 gene mutation. This dysfunction emerges during the development of the central nervous system and is associated with ID and ASD. Understanding of the pathophysiological mechanisms that underlie these neurodevelopmental disorders, particularly through the study of model organisms, is essential to improve therapeutic strategies.
These contents could be interesting :
- University François-Rabelais, Tours, France;
- Institut National de la Santé et de la Recherche Médicale, U930, Tours, France;
- Department of Molecular Biology, Radboud Institute for Molecular Life Sciences, Radboud University, Nijmegen, The Netherlands;
- PHENOMIN, Institut Clinique de la Souris (ICS), CNRS, INSERM, University of Strasbourg, Illkirch-Graffenstaden, France;
- Institut National de la Santé et de la Recherche Médicale, U966, Tours, France;
- Centre Hospitalier Régional Universitaire, Tours, France;
- Department of Cognitive Neuroscience, Department of Human Genetics, Radboudumc, Donders Institute for Brain, Cognition and Behaviour, Nijmegen, The Netherlands;
- Institut Cochin, 24 rue du Fg St Jacques, Paris, France;
- Institut National de la Santé et de la Recherche Médicale, U1016 Paris, France;
- Centre National de la Recherche Scientifique, UMR8104, Paris, France;
- University Paris Descartes, Paris, France;
- Centre National de la Recherche Scientifique, Institut de Pharmacologie Moléculaire et Cellulaire, Université Côte d’Azur, Valbonne, France;
- Department of Laboratory Medicine and Pathobiology, Faculty of Medicine, University of Toronto, Toronto, ON, Canada;
- Department of Human Genetics, Radboud University Medical Center, Nijmegen, The Netherlands;
- Radboud Institute for Molecular Life Sciences, Radboud University Medical Center, Nijmegen, The Netherlands;
- Molecular Neuropsychiatry & Development (MiND) Lab, Campbell Family Mental Health Research Institute, Centre for Addiction and Mental Health, Toronto, ON, Canada;
- Institut de Génétique et de Biologie Moléculaire et Cellulaire, University of Strasbourg Illkirch, France;
- Centre National de la Recherche Scientifique, UMR7104, Illkirch, France;
- Institut National de la Santé et de la Recherche Médicale, U964, Illkirch, France, IGBMC-CNRS UMR7104–INSERM U964, 1 rue Laurent Friès, BP 10142, Illkirch Cedex 67404, France