In the Web 2.0 and “Quantified Self” era, scientists are working to improve traditional data collection methods (face-to-face interviews, telephone interviews or paper questionnaires), which are expensive and time-consuming for study participants. Indeed, recruitment, monitoring and data collection tools need to be adapted to fit the habits of the younger generation. The Internet and smartphones offer numerous options for collecting data on participants’ lifestyle, environment and health, in a way involving few constraints, and that can even be fun. In May 2013, the INSERM team headed by Françoise Clavel-Chapelon (Unit 1018, Team 9 “Nutrition, Hormones and Women’s Health”) organized an international conference entitled “e-tools and social networks for epidemiology” in Paris. The main objective was to present academic research and ICT (Information and Communication Technology) innovations that can improve epidemiological research.
New data acquisition tools
Carol Boushey, from the University of Hawaii, presented her research on TADA (Technology Assisted Dietary Assessment) technology. TADA is a prototype application for smartphones that collects dietary information easily. The process is simple: you just need to take a picture of your plate before and after a meal. The program’s algorithms do the rest, detecting textures and volumes of foods, and reconstitute the exact composition and dietary intakes.

Carol Boushey demonstrating TADA (Technology Assisted Dietary Assessment)
New techniques to record data on physical activity were also presented. Stephen Intille (NortheasternUniversity, Boston) described
the work done by the Mobile Health Research Group on the use of smartphones and detectors to measure health and physical activity data.
Suzanne Garland (Royal Women’s Hospital, Melbourne) explained how the
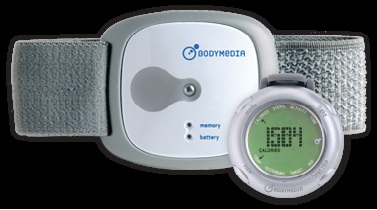
SenseWear® Armband is used in the Australian
Young Female Health Initiative study
. This electronic bracelet collects physiological data (skin temperature, heart rate, etc.) and provides instant information about the number of steps walked, the distance covered, calories burned, sleep cycle, etc.
Furthermore, geolocation techniques are increasingly finding applications in epidemiology. For example, the Veritas tool, presented by Yan Kestens (University of Montreal) based on online cards, can collect spatial information, and the tool presented by John Nuckols (ColoradoStateUniversity) combines recording of geolocation and environmental pollutants data.
Web 2.0 and e-Health
Several talks presented new recruitment techniques, networking, data management and loyalty card schemes run via Internet.
John Wark (Melbourne University) described how Facebook was used to recruit volunteers into the Young Female Health Initiative study.
Finally, several web platforms were presented, such as CleanWEB ePRO®, an integrated electronic management solution for clinical trials, various ad hoc e-health programs rolled out by the BePATIENT company, and the SenseDoc multisensor platform developed by the SPHERE research laboratory (Research Centre of the University of Montreal’s research centre), in collaboration with the RECORD Study team.

Le SenseWear® Armband de la société BodyMedia®
The major advantages of these web 2.0 platforms are the ability for participants to network, the collection of digital data that can be used very quickly, the connection to e-tools, sensors, smartphone applications that record information automatically, and the ability to obtain individual feedback based on the participant’s profile.
“In addition to saving time and to the improved reliability of data collected through these means, these resources have a fun and interactive component, which is a very important aspect to ensure continued participation of large cohort participants”, explains Françoise Clavel-Chapelon, INSERM Research Director and Principal Investigator of the E3N and E4N cohorts.
This first conference discussed how new tools such as social networks, e-health and spatial technology could have applications in the epidemiological studies of tomorrow. However, before using these innovative methods and tools in epidemiological studies, it is necessary to check their acceptability, reliability and validity. “The E4N cohort (see box below) will offer particularly favourable conditions to carry out this validation and standardization phase”, concludes Guy Fagherazzi, scientific manager of the E4N cohort.
The conference was organised by the INSERM team headed by Françoise Clavel-Chapelon (UMRS 1018, Team 9) who wanted to discuss new ways of collecting epidemiological data for the E4N cohort. The event was run jointly with AFCROs (Association française des CROs), in partnership with Aviesan Santé Publique, the INRIA and three competitive high-tech hubs in the Paris region (Cap Digital, Systematic Paris-Région and Medicen) brought together under the label of “TIC & Santé”. The conference received financial supports from the Mairie de Paris, the Université Paris-Sud and the Ligue contre le Cancer.
The E4N study is designed to be an extension of the E3N study by following-up the spouses, children and grandchildren of the E3N women. The objective is to better understand the onset of disease, from genetics, family environment and the environment outside the family. This project of a prospective family cohort has been selected by the “Investissements d’Avenir” programme from the ANR (Agence Nationale de la Recherche) for a follow-up over a nine-year period.
For more information: www.e4n.fr