Sorry, this press release is only available in French.
(French) Cancers de l’enfant – Nouvelle piste thérapeutique dans les tumeurs d’Ewing

Sorry, this press release is only available in French.
Sorry, this press release is only available in French.
Sorry, this press release is only available in French.

Sorry, this press release is only available in French.
Sorry, this press release is only available in French.
Since 2009, France has been running a major pilot clinical trial into liver cancer as part of the International Cancer Genome Consortium (ICGC) project. The aim of this project is to sequence the genomes of the tumors from several thousands of patients suffering from around fifty different cancers, in order to gain a better understanding of the part played by genetic alterations in the development of these cancers. The work carried out by Jessica Zucman-Rossi’s team (Inserm unit 674 “Functional genomics of solid tumors” working jointly with the Université Paris Descartes), revealed 4 genes that had never been described in liver tumors, even although they presented frequent alterations. These genes, identified as ARID1A, RPS6KA3, IRF2 and NFE2L2, are involved in important processes that lead to the development of tumors in the liver. This research was published in the May 6, 2012 issue of Nature Genetics.
The work done by the French teams is coordinated by the INCa and Inserm. It includes 4 programs that each study a particular type of breast cancer, aggressive prostate cancers, Ewing’s sarcoma and liver cancer.
© Inserm, Zucman-Rossi, Jessica
Liver cancer is the third most frequent cause of death in the world. It often occurs in persons whose liver is already affected by pathologies such as hepatitis B or C, excessive alcohol consumption, iron overload or obesity. These pathologies can lead to the development of cirrhosis of the liver.
In France, a pilot study was initiated in 2009 on a first series of 24 hepatocellular carcinomas within the framework of the ICGC sequencing consortium. The aim of this study led by Jessica Zucman-Rossi was to find new genes responsible for the development of liver tumors (tumor suppressor genes and/or oncongenic genes).
Using new genome sequencing techniques, the researchers were able to establish that the genetic code[1] of persons suffering from liver cancer often presented modifications, in which the G bases had been replaced by T bases. These mutations seem to be specific to and closely linked to liver cancers. This strongly suggests that, where there is no previous cirrhosis of the liver, a toxic agent is involved in the process and causes mutations in the DNA of these patients.
In tropical areas, compounds such as aflatoxin B1[2] are already well known for similar cancer-producing effects. New epidemiological and toxicological data will be needed to accurately determine what kind of genotoxic agents can affect patients living in France.
Identification of 4 new genes involved in liver tumors
An analysis of all the observed mutations brought to light four new genes that had never before been described in hepatic tumors, and yet they present recurrent genetic alterations: ARID1A, RPS6KA3, IRF2 and NFE2L2. In order to understand what part they play, these four genes were tested along with the 10 other genes, on samples from 125 hepatic tumors.
From a physiological point of view, it seems that certain of these genetic mutations seem to alter two signaling pathways known to the scientists: The WNT/p-Catenine pathway and the P53 pathway. Others on the other hand, are involved in triggering off oxidative stress, interferon or RAS signaling, thereby disturbing the state of the cells. Finally, in patients suffering from chronic alcohol intoxication, the chromosome remodeling genes (that stabilize DNA) are often altered, making them major factors that contribute to hepatic tumor genesis.
As far as Jessica Zucman-Rossi is concerned, “This study reveals new tumor-suppressor and oncogenic genes involved in hepatic carcinogenesis. New lines of investigation are still to be explored so that the future will see new medication that is able to target the genetic alteration and thus improve and tailor the treatment of the patients to treat the genome abnormalities identified in their tumors.”.
This work was coordinated and financed by the INCa as part of the Cancer Plan 2009-2013, in close collaboration with the Ligue nationale contre le cancer and Inserm.
The Institut national du cancer – The French partner of the internal ICGC consortium
The International Cancer Genome Consortium (ICGC) is a global program of high-level biomedical research. It is one of the most ambitious programs since the Human Genome project (https://www.icgc.org/). Launched in 2008, the ICGC program, that currently involves 14 countries, has the task of drawing up a complete description of the genomic, transcriptomic and epigenomic alterations of 50 cancer types or subtypes considered to be the most preoccupying both from clinical and society points of view. 25 000 cancer genomes are to be sequenced and analyzed and the results will gradually be entered into a database that will be accessible to researchers the world over. The list of genomic alterations specific to each type of cancer will enable us to develop functional investigations on the mechanisms of carcinogenesis, and will provide new strategies for prevention, diagnosis and treatment.
The member organizations and the research centers taking part in the ICGC have agreed to abide by standards governing informed consent and common ethical rules, in order to ensure that the identity of each participant is protected. The Institut national du cancer (INCa) has overall coordination of the French contribution to this program. It benefits from financing by INCa and Inserm amounting to 5 M € per year.
The ICGC program is an opportunity to consolidate the organized effort undertaken by the INCa in the field of high-level genomics. In 2012 INCa and Inserm took on 4 projects concerning liver cancer, HER2+ breast cancer, prostate cancer and Ewing’s sarcoma. A fifth program is to be started in 2012-2013
[1] based on the four ATCG bases
[2] Aflatoxine is a toxin that is secreted by a fungus(Ascomycete aspergillus flavus) that exists in large quantities on different seeds in hot, wet, conditions. It is a powerful oncogenic agent, affecting the liver in particular, and is also an ARNm sythesis inhibitor.
Sorry, this press release is only available in French.
The position of cellular nuclei in muscle fibres has an important role in some muscle weaknesses. Edgar Gomes, an Inserm researcher in the myology group at the Institute of Myology (mixed Inserm/UPMC unit) recently made this discovery in collaboration with an American team. The researchers identified several proteins involved in “correctly” positioning nuclei, which is required for the muscles to function. Their results are published in a letter in the Nature review, dated 18 March.
In order to move, living beings need muscles, and, more specifically, skeletal muscles that are controlled by the nervous system. Skeletal muscles are composed of cylindrical muscle fibres with a multitude of peripheral nuclei. Until now, little was known about the mechanism used to position nuclei on the edge of muscle fibres. A team of French-American researchers has tried to better understand the reasons behind nuclei layout.
Edgar Gomes and his team of collaborators have identified the mechanism involved in positioning nuclei in muscle fibres. The researchers identified (in Drosophila and mice) two proteins involved in positioning the nuclei: protein Kif5B, which belongs to the kinesin family (molecular motor), and protein MAP7, which is used to move different organelles (1) in cells.
This result was achieved by mutating MAP7 and Kif5b protein-coding genes in the Drosophila and by studying the development of the embryo. In this case, they observed that the nuclei were not correctly aligned in the muscle fibres.
“MAP7 is required to position nuclei in muscle fibre in Drosophila and in mammals” states Edgar Gomes, Inserm researcher. The research team succeeded in describing the nuclei-positioning mechanism in fibres, which involved the MAP7 protein and its interaction with the molecular motor: kinsin Kif5b. They demonstrated that a mutation of these proteins did not affect muscle extension or its attachment to the skeleton: only the position of the nuclei was affected.
By making both proteins interact together, Edgar Gomes’ team suggest that MAP7 binds with Kif5b to encourage nuclei positioning. “Furthermore, these proteins act together, both physically and genetically, and their physical bond is required for correct nuclei positioning. Our results show that they are required for the muscle to function correctly” underlines Edgar Gomes.
Muscular diseases lead to weaknesses in the fibres and can be associated with a cellular nuclei alignment failure. Edgar Gomes and his team have demonstrated that by correctly replacing the nuclei, the muscle recovers its functions. “We suggest that by correcting muscular positioning faults in patients suffering from myopathies, these patients may see improvements in their muscular functioning” concludes Edgar Gomes.
Footnote
(1) Specialized structures in the cell contained in the cytoplasm

A discovery presented by Catherine Seva, biologist at Inserm
Two research scientists, Catherine Seva and Audrey Ferrand, from a combined team of researchers from Inserm / Université Toulouse III – Paul Sabatier (Cancerology research center of Toulouse), have just developed a test to predict the risk of colorectal cancer. This test, based on the presence of a protein known as progastrine, can predict the risk of tumors occurring in patients who have already undergone surgery to remove polyps that were considered to be benign. In practice, these tests could be used for routine screening on resected polyps in order to identify patients presenting a high risk of developing precancerous lesions and for whom no monitoring is recommended at present. The results of this work are published in the review Cancer prevention research.
Bowel cancer is the 4th highest cause of death from cancer in France. So March 2012 has been named “Blue March”, colorectal cancer awareness month.
A predictive test to assess the risks of bowel cancer
Colorectal (or bowel) cancer is the second most frequent cancer. Hyperplasic polyps are the most frequently occurring colorectal lesions Nearly one quarter of the European population between the ages of 20 and 54 will develop this type of polyp. For a long time, these lesions were considered to be benign, so currently there is no recommended monitoring of these patients once the polyps have been removed surgically. However, some of these polyps could be precursory signs of bowel cancers. Up until now, we had no way of identifying the sub-group of polyps with potential for malignancy.
So with the aim of finding a biomarker that could predict the risk of bowel cancer in patients presenting with hyperplasic polyps – the most benign type – Catherine Seva and her co-workers carried out a 10-year retrospective clinical study. They analyzed the presence of a particular protein name progastrine in hyperplasic polyps from 74 patients. Progastrine is already known to play a part in colic cancerogenesis. This protein, produced by tumerous colorectal cells, is not present in healthy bowel cells. So the researchers tried to find out whether the expression of this protein could predict the occurrence of cancerous lesions in the years following surgical resection of the polyps. “When we first put forward this hypothesis, we had in mind the idea that this was a step towards ensuring suitable monitoring and very early detection of colorectal cancer”, explains Catherine Seva, Research Director at Inserm.
Thanks to these analyses, the research scientists showed that there was indeed a significant relationship between high levels of progastrine and future occurrence of precancerous lesions. And yet although these polyps had been considered as benign and risk-free, within a period of 2 to 10 years, 100% of patients who presented high levels of progastrine developed adenomas, renowned as early colorectal cancer lesions. Inversely, in patients who expressed little or no progastrine, no lesion developed at all over the 10 years following the removal of their polyps.
Based on these results, the researchers compiled a predictive test based on the age of the patient and prograstrine immunohistochemical marking. This highly sensitive and specific tests can predict tumors in patients who have developed hyperplasic polyps.
“There is no recommended monitoring at the present time, and yet measuring the prograstrine expression in hyperplasic polyps could identify the population of patients who have a high risk of developing a precancerous lesion”, concludes Audrey Ferrand, research scientists at Inserm and signatory of this paper.
As a result of this study, it could be envisaged to include a larger number of patients to valid this as a routine test.
This research has been patent-protected by Inserm Transfert.
A joint Institut Curie/Inserm team headed by Dr. Olivier Delattre has discovered the existence of a new form of bone sarcoma, affecting mainly adolescents and young adults.
Although similarities exist with Ewing’s tumor in the clinical field, a different chromosomal abnormality is responsible for the development of this sarcoma.
This new information should enable to improve patient management.
The results of this discovery have been published online in Nature Genetics on March 4.
Institut Curie is the French center of excellence for the detection and clinical management of Ewing’s tumors, performing the majority of molecular diagnostic tests for this pathology. The team of Olivier Delattre, research director of Inserm 830/Institut Curie “Genetics and Biology of Cancers” unit, was thus able to examine the genetic material of 594 children suspected to have Ewing’s tumor. Using a new sequencing technology called Next generation sequencing (NGS), they were able to analyze large volumes of genetic material simultaneously. During this study, the researchers discovered a different mutation than the one responsible for Ewing’s tumor, in some samples[1].
They managed to isolate 24 samples with this new mutation: an inversion of the X chromosome resulting in the fusion between two neighboring genes, BCOR and CCNB3. The very small distance separating these genes, which are located on the same chromosome, made them impossible to discover until now with the traditional diagnosis methods.
Then the researchers compared the gene expression profiles of the samples with those of Ewing’s tumor samples. The “ID card” of the new form of sarcoma indicates a different expression for approximately 3,000 genes. “This ‘new’ fusion is certainly responsible for a different sarcoma rather than a variant of Ewing’s tumor”, declared Dr. Olivier Delattre.
“It is the first time that the possibility of identifying a new tumor entity using high throughput sequencing tools is established”, added Inserm researcher Franck Tirode, co-author of the published study. He then noted: “In a single study, we have detected 24 positive cases for this translocation, and that is a significant number”. “In absolute value, these 24 cases are equivalent to the number of cases due to rare translocations of Ewing’s tumor”, added co-author Gaëlle Pierron, coordinator of Institut Curie’s Somatic Genetics unit.
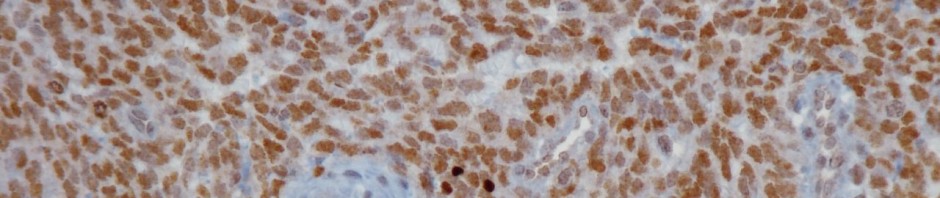
Presence of the fusion protein was verified on tumor samples using immunohistochemistry techniques that enable to locate specific proteins. “Detection of the over-expression of the CCNB3 protein, because of its fusion with BCOR, is extremely specific to these tumors, enabling a simple diagnostic test”, explained Gaëlle Pierron.
Towards improved patient management
“Currently, patients are treated similarly to patients with Ewing’s tumors. It will now be possible to carry out clinical studies to look for the possibility of other treatment methods”, declared Olivier Delattre.
It will be possible to detect other patients affected by this new type of sarcoma. Identification of the fusion between the two genes could enable diagnosis of this new form of tumor in new patients, who will then benefit from adapted treatment.
“Actual advances in oncology result from the dialogue between physicians and researchers. It is important to provide everyone with the means to strengthen this exchange”, concluded Dr. Olivier Delattre.
Researchers will now work to decipher the active mechanism of the fusion protein that results in the formation of the tumor. In particular, they will try to understand why this new type of sarcoma is clinically similar to Ewing’s tumor, while the tumors are not biologically similar. Common activation of specific intracellular signaling pathways could be the cause.
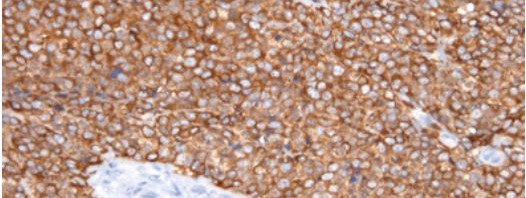
Sections of bone sarcoma with marking of the CCNB3 fusion protein
Right: Section of the newly identified tumor. The brown staining shows the expression of the BCOR-CCNB3 fusion protein in cells.
In addition to Inserm and Institut Curie, this research was financed by the French League Against Cancer and by the French National Cancer Institute.
In addition, Dr. Olivier Delattre’s team also receives financial support from APAESIC (Association of Parents and Friends of Children Treated at Institut Curie), Les Bagouz à Manon, Pas du Géant, Olivier Chape, Les Amis de Claire and Courir pour Mathieu, as well as the Fédération Enfants et Santé.
[1] Ewing’s tumor is characterized by an accidental exchange of genetic material between two chromosomes, resulting in the formation of a mutated gene producing an abnormal protein called EWS/FLI-1.
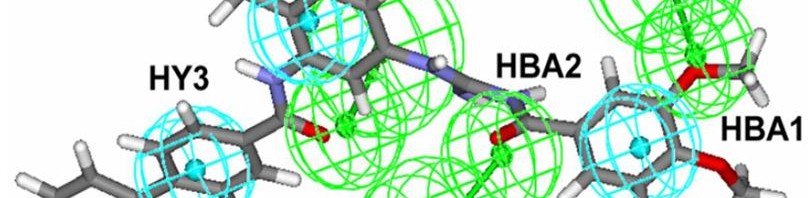
A French-Italian team headed by researchers from CNRS and Inserm (1) has recently discovered a new family of compounds that could make it possible to treat numerous cancers, particularly brain tumours and skin cancers. These substances, patented by CNRS, block a chain of molecular reactions known as the Hedgehog signalling pathway, the disruption of which could be involved in several cancers. These compounds could become new medicines in the long term. In the short term, they should prove to be precious tools in elucidating the role of the Hedgehog pathway in the development of these tumours and their resistance to treatment. This work, carried out in collaboration with the Laboratoire d’Innovations Thérapeutiques (CNRS / Université de Strasbourg), is published in the Journal of Medicinal Chemistry.
The Hedgehog signalling pathway is a cascade of complex biochemical reactions. Very active during embryogenesis, it participates in cell proliferation and differentiation, as well as in the development of numerous tissues. In adults, it plays a key role, particularly in maintaining stem cells in the brain. The disruption of this pathway could participate in the development of many cancers, especially very aggressive brain tumours in children.
Malfunctions affecting the Hedgehog pathway can be caused by mutations of a membrane receptor known as Smoothened, an essential link permitting the activation of the Hedgehog pathway. Several pharmaceutical laboratories have developed compounds capable of blocking Smoothened. Thanks to these receptor antagonistic compounds (2), they have succeeded in stopping the development of certain tumours. However, experiments conducted on animal models and in humans demonstrate the onset of resistance to these treatments. New mutations of Smoothened in the tumoral cells annihilate the effects of the antagonists responsible for inactivating it. For this reason, it is important to find new antagonists and to better understand the mechanisms underlying this type of resistance.
In order to discover new Smoothened antagonists, the team of researchers coordinated by Martial Ruat adopted a novel strategy, namely the virtual screening of computerized molecular databases.
Among the 500,000 or so molecules included in these databases, they searched for those whose structure could produce the same effect as molecules already known to inhibit Smoothened. The scientists selected one molecule out of about 20 candidates. By slightly modifying its structure in order to optimize it, they discovered a family of compounds known as MRT and tested their biological activity on cultured mouse cells.The team discovered that MRT compounds, and more particularly acylguanidine MRT83, block the proliferation of cells suspected of causing brain tumours. In addition, these new compounds inhibit Smoothened with an activity similar to or exceeding that of already known compounds.
Several years of tests will be necessary before promising new substances such as MRT compounds can be marketed as medicines. Nevertheless, their properties could shed new light on the functioning, the three dimensional structure and the location of Smoothened receptors. These MRT compounds could contribute to understanding the cause of tumour resistance. This study could lead to the discovery of new targets and therapeutic strategies for combating certain cancers.
(1) In the ‘Neurobiology & development’ research unit (CNRS), in collaboration with the University of Sienna (Italy)
(2) When bound to the same cell receptors as one other substance, an antagonist substance blocks all or part of the effects normally produced by the cell. Here the receptor antagonist has an ‘inverse’ effect compared with that of the ‘muted’ receptor.