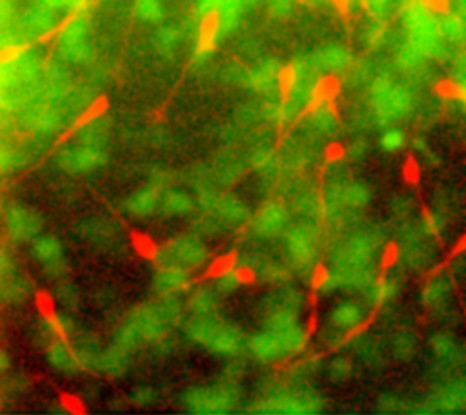
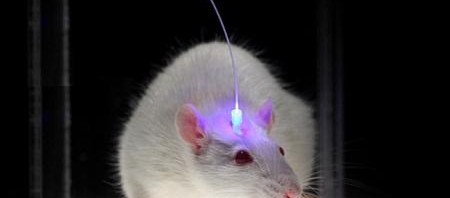
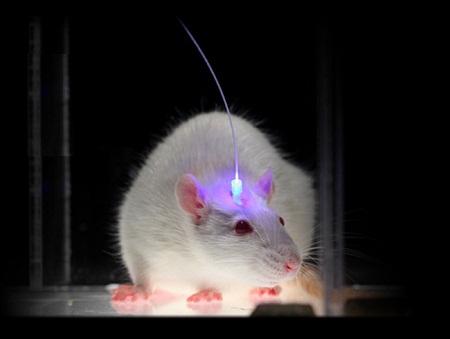
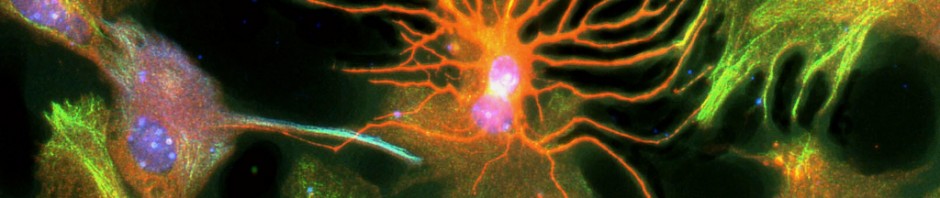
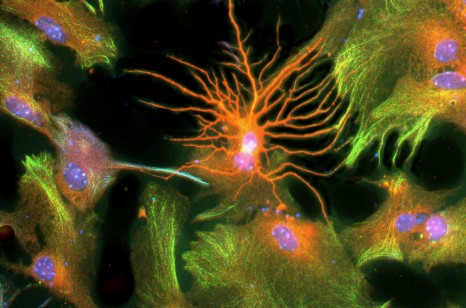
A team of researchers at Inserm led by Cyril Herry (Inserm Unit 862, “Neurocentre Magendie,” Bordeaux) has just shown that interneurons located in the forebrain at the level of the prefrontal cortex are heavily involved in the control of fear responses. Using an approach combining in vivo recordings and optogenetic manipulations in mice, the researchers succeeded in showing that the inhibition of parvalbumin-expressing prefrontal interneurons triggers a chain reaction resulting in fear behaviour. Conversely, activation of these parvalbumin interneurons significantly reduces fear responses in rodents.
This research is published in the journal Nature
Some traumatic events may lead to the development of severe medical conditions such as anxiety disorders or posttraumatic stress disorder (PTSD).
Anxiety disorders have a prevalence of approximately 18% worldwide.
Despite successful treatments, some patients relapse, and the original symptoms reappear over time (fear of crowds, recurring nightmares, etc.). An understanding of the neuronal structures and mechanisms involved in this spontaneous recovery of traumatic responses is essential.
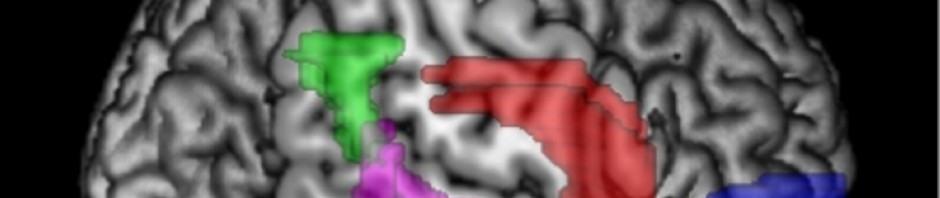
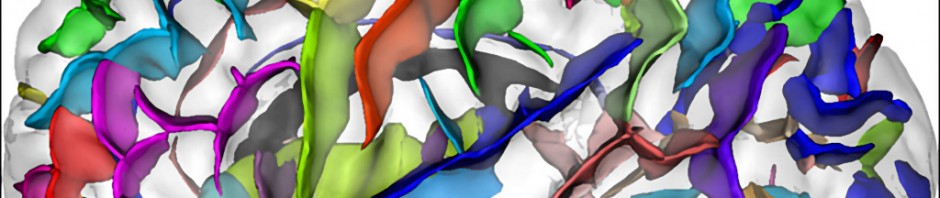
All observations made by researchers indicate that fear behaviours are controlled in the forebrain at the level of the dorsomedial prefrontal cortex. This control of fear behaviour is based on the activation of neurons in the prefrontal cortex that are in contact with specific areas of the amygdala.
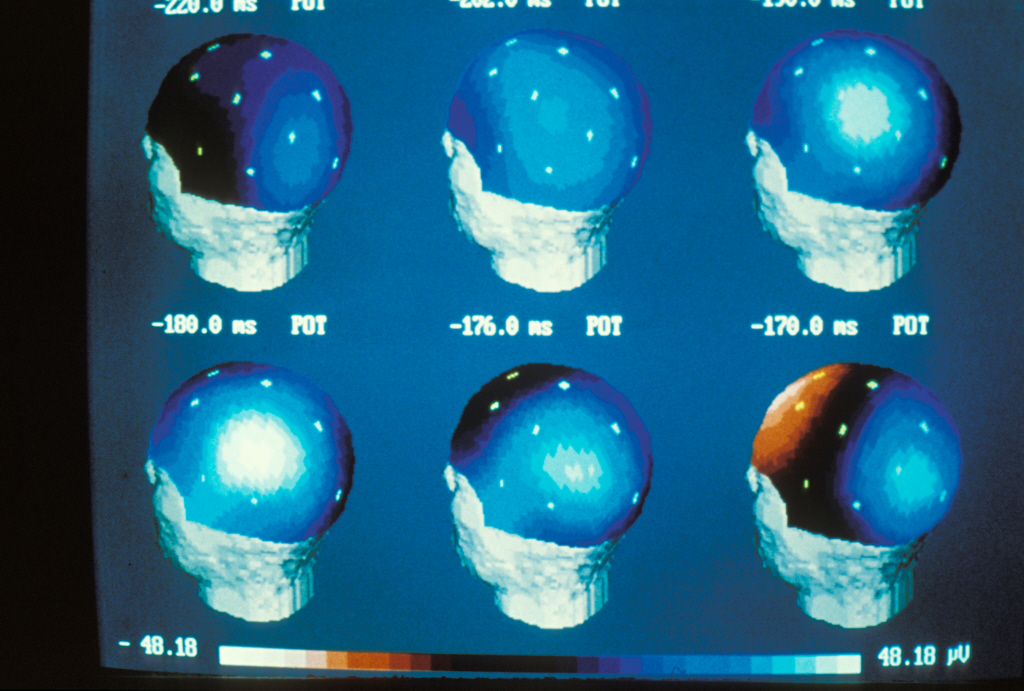
Using an innovative approach combining electrophysiological recording techniques, optogenetic manipulations and behavioural approaches, the researchers were able to demonstrate that fear expression is related to the inhibition of highly specific interneurons—the parvalbumin-expressing prefrontal interneurons.
More specifically, inhibition of their activity disinhibits the activity of the prefrontal projection neurons, and synchronises their action.
Synchronisation of the activity of different neuronal networks in the brain is a fundamental process in the transmission of detailed information and the triggering of appropriate behavioural responses. Although this synchronisation had been demonstrated as crucial to sensory, motor and cognitive processes, it had not yet been examined in relation to the circuits involved in controlling emotional behaviour.
“Our results identify two complementary neuronal mechanisms mediated by these specific interneurons, which accurately coordinate and increase the neuronal activity of prefrontal projection neurons, leading to fear expression,” explains Cyril Herry.
The identification and better understanding of these neuronal circuits controlling fear behaviour should allow the development of new treatment strategies for conditions such as posttraumatic stress disorder and anxiety disorders. “We could, for example, imagine the development of individual markers for these specific neurons, or the use of transmagnetic stimulation approaches to act directly on excitatory or inhibitory cells and reverse the phenomena.”
How is fear analysed in an animal?
From an experimental standpoint, the classic Pavlovian conditioning procedure involves associating one stimulus, such as a sound, with another, unpleasant stimulus, such as a small electric shock. This first step allows the animal to establish a persistent aversive memory. In other words, the animal comes to remember and learn that the sound is associated with an unpleasant state, and an immobility response is routinely triggered, which is a good indication of fear in an animal.
In the second step, the extinction procedure involves repeated presentation of the sound alone, inducing a temporary inhibition of the conditioned fear responses. This inhibition is only temporary, as the mere passage of time favours the spontaneous recovery of the conditioned fear responses, which, from the clinical standpoint, may be associated with the phenomenon of relapse into traumatic responses seen following the treatment of posttraumatic stress disorder using exposure-based therapies.