The scientific community agrees that autism has its origins in early life—foetal and/or postnatal. The team led by Yehezkel Ben-Ari, Inserm Emeritus Research Director at the Mediterranean Institute of Neurobiology (INMED), has made a breakthrough in the understanding of the disorder. In an article published in Science, the researchers demonstrate that chloride levels are elevated in the neurons of mice used in an animal model of autism, and remain at abnormal levels from birth. These results corroborate the success obtained with the diuretic treatment tested on autistic children by the researchers and clinicians in 2012, and suggest that administration of diuretics to mice before birth corrects the deficits in the offspring. They also show that oxytocin, the birth hormone, brings about a decrease in chloride level during birth, which controls the expression of the autistic syndrome.
This work is due to appear in the 6 February 2014 issue of Science.
Neurons contain high levels of chloride throughout the entire embryonic phase. As a result, GABA, the main chemical messenger of the brain, excites the neurons during this phase instead of inhibiting them, in order to facilitate construction of the brain. Subsequently, a natural reduction in chloride levels allows GABA to exercise its inhibitory role and regulate the activity of the adolescent/adult brain. In many brain disorders (childhood epilepsy, cranial trauma, etc.), studies have shown abnormally high chloride levels. Having made various observations, Dr Lemonnier’s team (Brest), and Yehezkel Ben-Ari’s team at Inserm carried out a clinical trial in 2012, based on the hypothesis of high chloride levels in the neurons of patients with autism. The researchers showed that administration of a diuretic to children with autism (which reduces neuronal chloride levels) has beneficial effects[1]. The results of the trial supported this hypothesis, but because high neuronal chloride levels could not be demonstrated in children with autism, it was not possible to prove the mechanism proposed or justify the treatment.
In the present study, the researchers therefore used two animal models of autism, a genetic model, Fragile X syndrome, which is the genetic mutation most frequently associated with autism, and another, generated by injecting the spleen of pregnant mice with sodium valproate, a product known to generate abnormalities in children, including autistic spectrum disorder.
A high level of chloride in the brain
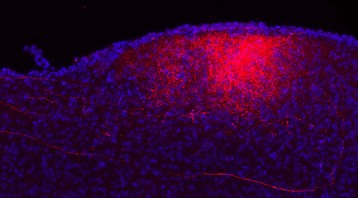
For the first time, the researchers recorded the activity of neurons at the embryonic stage and immediately after birth in order to observe modifications in chloride levels. These observations showed that neuronal chloride levels are abnormally high in both young and adult animals used in the autism model. GABA strongly excites neurons, and the researchers recorded aberrant electrical activities in the brain, which persist in adult animals.
The fall in chloride level, a particularly impressive phenomenon seen at birth in control animals, is absent in both of these animal models, and the neurons have the same chloride level before and after birth. These high levels are due to reduced activity of a chloride transporter, thus preventing transport of chloride out of the neuron. As a result, a major feature of neurons during birth is abolished in animal models of autism.
“Chloride levels during delivery are determinants of the occurrence of autism spectrum disorder,”
explains Yehezkel Ben-Ari, an Emeritus Research Director at Inserm.
Beneficial effects of the diuretic on brain activity
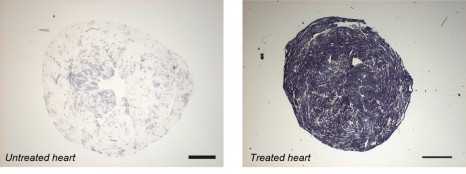
The researchers administered a diuretic treatment to the mother (in both animal models) for 24 h shortly before delivery to see if this would restore brain inhibition in the offspring. They showed that the drop in chloride level was re-established in the neurons several weeks after a single treatment during birth. According to the research team, antenatal treatment restored brain activity to approximately normal levels, and corrected the “autistic” behaviour in the animals once they became adults.
“These results thus validate the working hypothesis that led us to the treatment we developed in 2012,” states the principal author of the study.
Oxytocin, the birth hormone, naturally reduces chloride levels
The role of oxytocin in reducing neuronal chloride was also studied. The researchers had previously shown in 2006[2] that this hormone, which triggers labour, also has many beneficial actions on the brains of newborns, including protective effects in the event of complications during delivery, and even analgesic properties. Oxytocin acts like the diuretic, reducing the intracellular chloride levels.
In the present study, the team tested the long-term effects of blocking the actions of the hormone before birth. A drug that blocks the signals generated by oxytocin was injected into pregnant mice. The researchers evaluated the effects of this blockage on the offspring, and showed that it reproduced the entire autism-like syndrome in them, both with respect to the electrical and behavioural aspects (identical to the two animal models of autism). As a result, the hormone’s natural actions, just like those of the diuretic, are crucial to this delicate phase, and may control the pathogenesis of autism via the cellular chloride levels.
“These data validate our treatment strategy, and suggest that oxytocin, by acting on the chloride levels during delivery modulates/controls the expression of autism spectrum disorder,”
states Yehezkel Ben-Ari.
Taken together, these observations suggest that earliest possible treatment is essential for maximum possible prevention of the disorder.
This work raises the importance of carrying out early epidemiological studies in order to better understand the pathogenesis of the disorder, especially through analysing data on deliveries where a drop in chloride has occurred. Indeed, complicated deliveries with episodes of prolonged lack of oxygen, for example, or complications during pregnancy, such as viral infections, are often suggested as risk factors.
Finally, given the role of oxytocin in triggering labour, “although it is true that epidemiological data suggesting that scheduled caesarean deliveries may have increased the incidence of autism are controversial, it nonetheless remains that these studies should be followed up and extended in order to confirm or refute this relationship, which is still possible,” insists Yehezkel Ben-Ari, who concludes, “To treat this type of disorder, it is necessary to understand how the brain develops and how genetic mutations and environmental insults modulate brain activity in utero.”