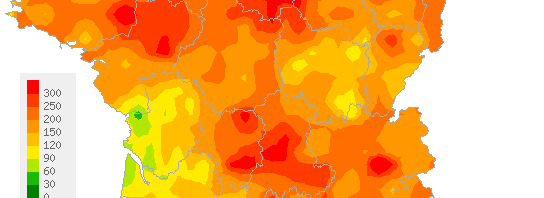
In Guinea, the epidemic caused by Ebola infection is still in the active phase nearly one year after it began. It is essential that measures for surveillance, control and management be accompanied by study of innovative therapies in order to reduce mortality. Potentially useful drugs include favipiravir (T-705), an antiviral drug already tested against influenza virus in adult humans (and well tolerated). The latter (no more than other potential treatments) has never been tested in humans for treating Ebola haemorrhagic fever, but its efficacy has been demonstrated in vitro and in mice.The pilot clinical trial conducted by Inserm is aimed at testing the efficacy of favipiravir in reducing mortality in individuals infected by Ebola virus in Guinea. It began on tuesday 23rd december 2014 in Gueckedou.
- What does this clinical trial involve?
The trial conducted by Inserm is a non-randomised open label multicentre Phase IIb trial All patients will receive the drug for 10 days. The drug favipiravir, which comes in the form of a 200 mg scored tablet (the tablet can be crushed and dissolved in a drink), will be administered according to the following dose regimen :
– Adults: D0: 2,400 mg at H0, 2,400 mg at H8 and 1,200 mg at H16, then 1,200 mg twice a day for 9 days;
– Children: doses adjusted to body weight.
Details of this dose regimen are the subject of a publication in the journal The Lancet Infectious Diseases.
All patients entering these various centres at the time of the trial may take the drug favipiravir if they so wish. The main analysis of the trial will be focused on patients taking the treatment at disease onset (within 48 h of the start of symptoms), based on the hypothesis that the drug is most active when administered early. However, all patients capable of swallowing the drug will be offered the treatment, regardless of how long they have had symptoms when they arrive.
Before being enrolled in the trial, each participant will give his/her consent. Consent will indicate that they have fully understood the clinical protocol. The latter will be explained prior to consent, in the appropriate language, by professionals in psychosocial counselling.
Since the symptoms are somewhat non-specific (fever, diarrhoea, vomiting), the researchers will perform PCR (a test based on gene amplification), which must be positive for Ebola virus before the treatment is offered.
In order for the results to be interpretable, the researchers must enrol 60 people responding to these criteria, something that may result in their treating several hundred people in total, for all durations of symptoms combined. Interim analyses will be conducted on every 20th patient treated to “survey” the safety and efficacy of the treatment, and to decide on the relevance of continuing the trial in the exact same way, or with modifications.
● Where will this trial take place?
The trial will take place in three Ebola Treatment Units (ETU) in Guinea:
– Gueckedou (managed by MSF),
– Macenta (managed by the French Red Cross),
– Nzérékoré (managed by the Alliance for International Medical Action, ALIMA).
The team conducting the clinical trial is based in Guekedou, in the Ebola Treatment Unit established by Doctors Without Borders/Médecins Sans Frontières (MSF). It should comprise some 30 individuals in total, including researchers, healthcare professionals and anthropologists. Coordination will be provided by French researchers from Inserm, and Guinean researchers from the Centre for Education and Research in Rural Health, under the authority of the National Ebola Response Coordination Committee in Guinea.
Relations with the community will be a central concern for the researchers, given the background of a serious epidemic and major health crisis, combined with a climate of justifiable distrust of outside interventions among the populations. A community committee to monitor the trial will be put in place before the trial is launched.
Coordinating investigators: Prof. Denis Malvy and Dr Sakoba Keita
Lead scientists: Dr Xavier Anglaret and Prof. France Mentré
● Provisional schedule
Enrolment is planned between 15 December 2014 and 15 March 2015.
Duration of involvement by each participant: 30 days
Total expected duration of the protocol: 4 months
Total duration of the project: 8 months
The first results will be published in March 2015 at the earliest.
The European Commission is funding this trial for an amount of €700,000.
Frequently asked questions:
Why not administer the drug vs a placebo?
For a disease with a very high risk of mortality, the evidence that a treatment is effective can be deemed convincing on observing a lower mortality than expected, without recourse to a placebo-controlled comparative trial. The principles of the experimental design for this trial are those retained for all the other trials being arranged.
Can there be side-effects from the treatment, and how are they monitored?
Favipiravir has been well tolerated in trials where it was used against influenza. Since the dose employed against Ebola is higher, side-effects will, however, be closely monitored, although problems of toxicity are not generally expected.
And if the results are inconclusive?
Depending on the first interim results, the trial can be interrupted in order to move on to another drug, or continued, either on its own or in combination with other antiviral drugs or other interventions.
What are the terms of the contract with the Japanese company that sells favipiravir?
The terms of this contract are confidential, but Inserm’s private subsidiary, Inserm Transfert, will safeguard Inserm’s interests and those of the populations.
What ethical questions are being raised by this trial?
This trial is being conducted under particularly difficult conditions, but ethical considerations are making us face these conditions in order to make progress, rather than do nothing. Populations affected by Ebola, like any population, are entitled to have promising therapeutic avenues explored using rigorous methods.