A team of French researchers from the CEA, AP-HP, CNRS, Inserm, Inra, ENVT, Institut Pasteur and MacoPharma have demonstrated that multiple variants of prions can coexist and manifest themselves in different clinical forms depending on the conditions of transmission. The results of this study are published in Nature Communications, dated 2 November 2017.
Fewer than 250 cases of variant Creutzfeldt-Jakob disease (vCJD) have been identified in the world, mainly in the United Kingdom, following the exposure of human populations to Bovine Spongiform Encephalopathy (BSE, or ‘mad cow disease’). However, over 99% of the human infections by these prions of bovine origin could remain silent according to British studies carried out on samples of appendices, which suggests a risk of inter-human transmission linked to such apparently healthy carriers, in particular through blood transfusions.
Assessment of the transfusional risk
Within the scope of the assessment of the transfusional risk linked to vCJD, researchers from the CEA, La Pitié-Salpêtrière university hospital, AP-HP, CNRS, Inserm, Inra, ENVT, Institut Pasteur and MacoPharma[1] exposed mice and macaques to blood products from vCJD-infected donors.
Through techniques traditionally used for the diagnosis of prion diseases (histology, biochemistry), the researchers found few cases of transmission of vCJD in its classic form (less than 3% of the animals exposed). These cases were mainly observed when the donors were strongly infected, which corresponds to the situation observed in humans[2].
Observation of myelopathies
On the other hand Emmanuel Comoy, CEA researcher and principal author, specifies, “in a significantly higher proportion (two to five times more according to the models studied, i.e. almost 10% of the animals exposed), some recipient animals developed fatal neurological syndromes different from vCJD”.
These syndromes do not present all of the classic characteristics of prion diseases (vacuole formation in the brain and accumulation of an abnormal form of the prion protein[3] resistant to deterioration by enzymes). They are referred to as myelopathies in affected primates because they are mainly centred on the spinal cord. These syndromes show themselves to belong to prion diseases, since their retransmission to the mice leads to the appearance of the classic characteristics of prion diseases.
Infection by healthy carriers
The myelopathic form of the disease was observed in animals transfused with blood from apparently healthy donors, i.e. donors exposed to prions and without an identifiable pathology, as well as from blood from patients presenting the clinical signs of vCJD.
The study thereby reveals a diversity of syndromes, from the healthy carriers to the typical form of vCJD: multiple variants of prions could therefore coexist and emerge under different clinical forms depending on the conditions of transmission (route of exposure, dose, nature of the contaminating product, aggregated or dispersed state of the prions).
In a context where human exposure through food intake to the BSE agent could have resulted in the emergence of numerous, apparently healthy, carriers, the researchers note that their study – which challenges the unicity of the vCJD/BSE origin – underlines the difficulty in detecting and preventing the transmission of all forms of prion infections. These questions are currently the subject of additiona.
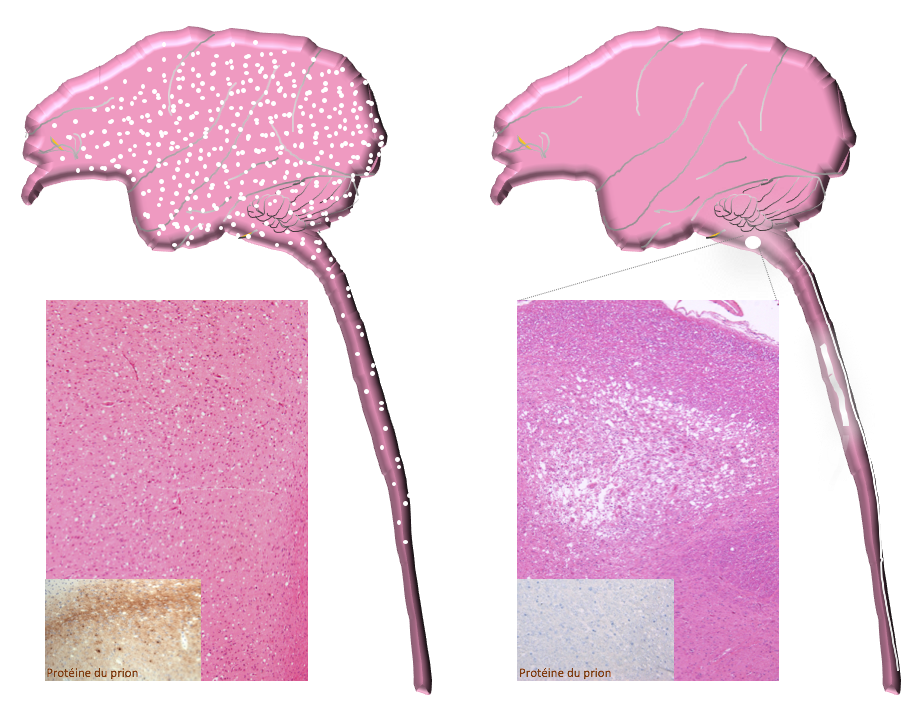
Comparison of the lesions observed: The macaques developing a vCJD present a vacuolation and an accumulation of the abnormal prion protein in the entire nervous system (figure on left), whereas the lesions in the myelopathic macaques are concentrated in the lower cervical spinal cord and the brain stem, with no accumulation of abnormal prion protein detectable using classic techniques (figure on right). © CEA/E. Comoy.
[1] Institut François Jacob (CEA), Brain and spinal cord institute (CNRS/Inserm/UPMC/La Pitié-Salpêtrière university hospital, AP-HP), Neuromyogenics institute (CNRS/Inserm/Univ. Cl. Bernard Lyon 1/Lyon Civil Hospices), Host-pathogen Interactions laboratory (Inra/ENVT), Macopharma, Biology of Infections unit (Institut Pasteur), laboratory of Neurodegenerative diseases: mechanisms, therapies, imaging (CNRS/CEA/Univ. Paris Sud).
[2] Over the last ten years, four cases of contamination by the vCJD agent in the United Kingdom have been attributed to the transfusion of non-deleukocyted blood products (so-called ‘whole’ blood, whose white cells have not been removed) coming from donors who themselves developed a vCJD shortly after their blood donation. At present, blood products are systematically deleukocyted.
[3] A pathological prion is a pathogenic agent made up of a protein whose conformation or withdrawal is abnormal and which, unlike infectious agents such as viruses or bacteria, or even parasites, has no nucleic acid (DNA or RNA) as a medium for infectious information.