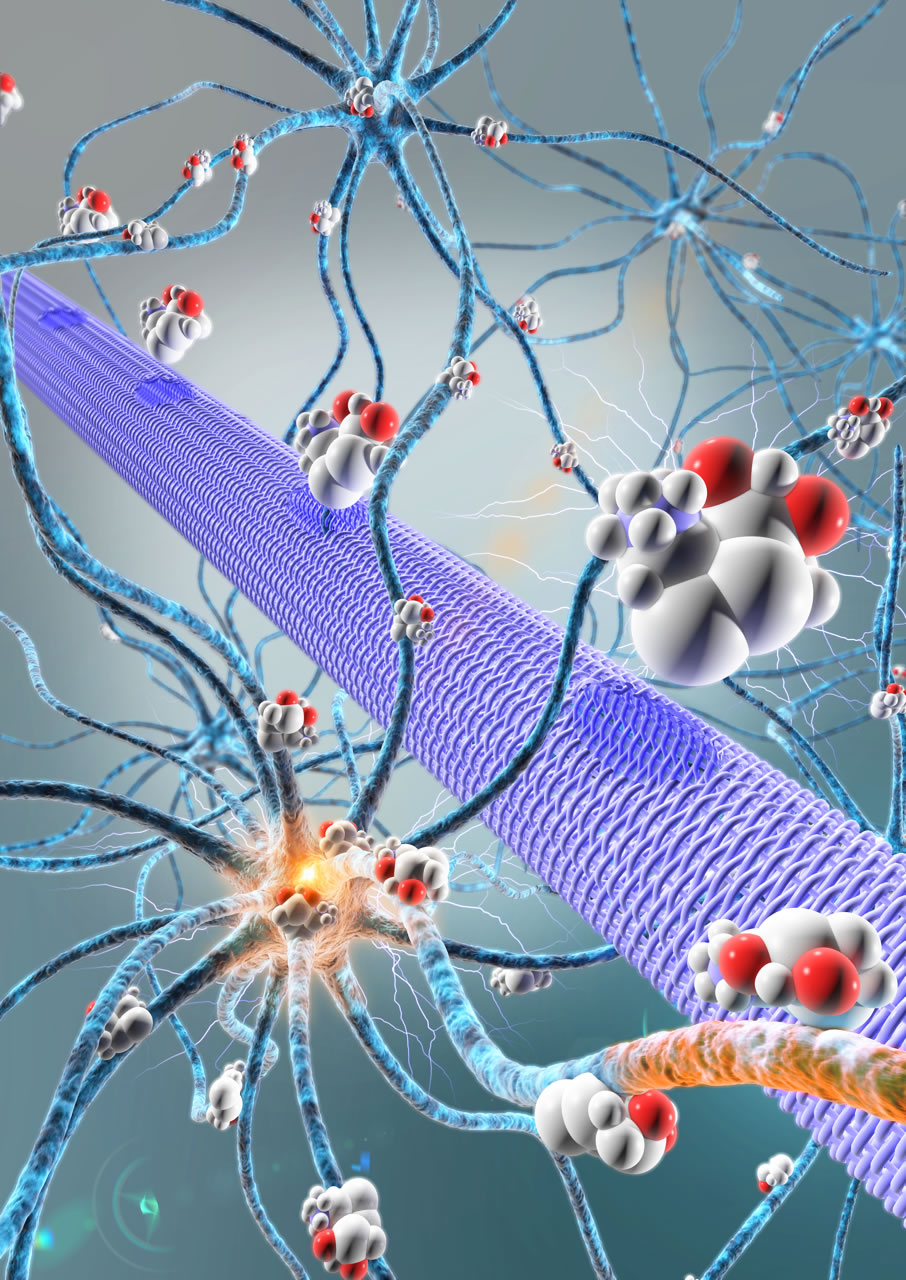
Scientists have just identified in the mouse, and then confirmed in humans, a new factor that regulates addiction. Glutamate, a neurotransmitter[1], contributes to regulating dopamine release in the nucleus accumbens, one of the cerebral structures of the reward system. More precisely, it is its subtle balance with another neurotransmitter – acetylcholine – that prevents up-regulation of the system and entry into addiction. This discovery, which opens up new therapeutic perspectives, was made by neurobiologists in the Neurosciences Paris-Seine laboratory (Institut de Biologie Paris-Seine, CNRS/Inserm/UPMC) and the Douglas Mental Health University Institute (McGill University, Montreal, Canada), working in association with human genetics specialists at Institut Mondor de Recherche Biomédicale (Inserm/UPEC). Their work was published on 4 August 2015 in Molecular Psychiatry.
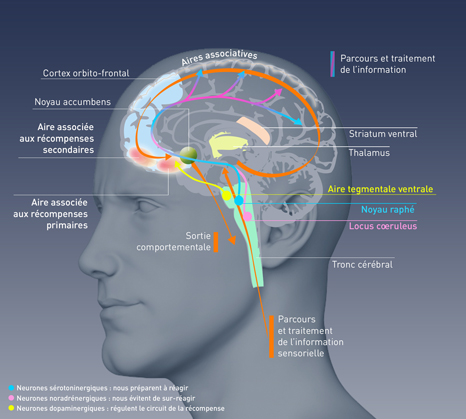
In the context of drug taking, dopamine levels rise in the cerebral structures that form the reward system. The intensity and rapidity of dopamine release provide a basis for the processes that will lead to the development of addiction. The cholinergic neurons in the nucleus accumbens, one of the centers of reward, are known to regulate this dopamine release. While most neurons only release a single neurotransmitter, a French-Canadian team led by CNRS researcher Salah El Mestikawy showed in 2002 that these acetylcholine-using neurons are also able to utilize glutamate. These neurons, which are to some extent “bilingual”, can thus both activate (via acetylcholine) and inhibit (via glutamate) dopamine secretion.
In this new study, much of it carried out by Diana Yae Sakae for her thesis project supervised by Salah El Mestikawy, the scientists showed that when they inhibited a gene essential to this signaling by glutamate (called VGLUT3) in mice, the animals became more vulnerable to cocaine. They experienced enhanced stimulant effects of the drug, developing “addiction” more easily and being more likely to “relapse” after a period of abstinence. The glutamate from these acetylcholine neurons therefore plays an important regulatory role in limiting cocaine addiction.
The scientists then wanted to determine whether this mechanism also applied to humans. In cocaine and/or opiate-dependent adults, they looked for mutations of the gene that had rendered the mice “addicted”. At the Institut Mondor de Recherche Biomédicale, Stéphane Jamain’s team observed that a mutation of this gene was ten times more common among severely dependent patients than in individuals without psychiatric symptoms. This mutation may explain the greater vulnerability to addiction of these patients[2]. In any case, these observations appear to confirm the role of glutamate in the addictive mechanism.
This work has thus clarified the neuronal mechanisms that underlie the search for hedonic sensations: contrary to what scientists thought until now, these findings show that it is not acetylcholine alone that regulates dopamine release, but a balance between acetylcholine and glutamate.
At the same time, the scientists identified an unsuspected target for the treatment of drug addiction. Indeed, although acetylcholine has numerous other functions in the brain and muscles, this glutamate signaling is more specific. The next step is to identify the receptor involved so that pharmacological therapies can be developed.[1] In order to communicate, neurons use chemical substances called neurotransmitters. Conventional neurotransmitters include dopamine, serotonin, acetylcholine and glutamate, etc.
[2] That said, even within the group of severely dependent patients this mutation was only present in 5% of cases, indicative of the multifactorial nature of addiction and more generally the complexity of psychiatric diseases



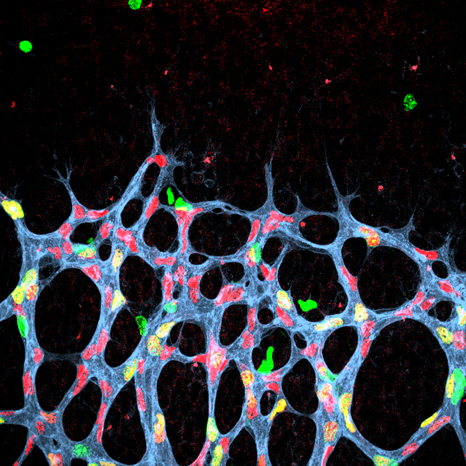
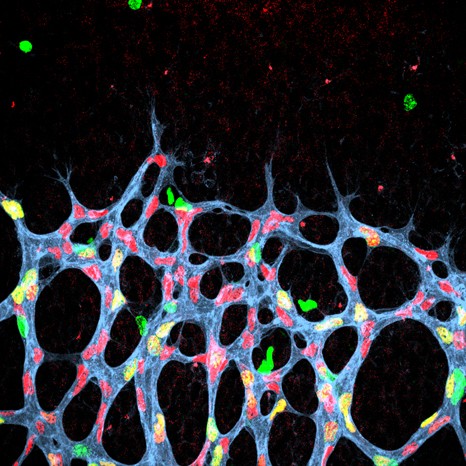
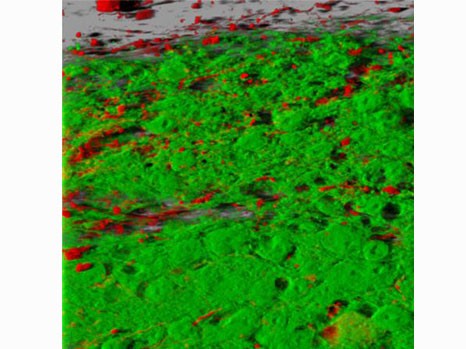
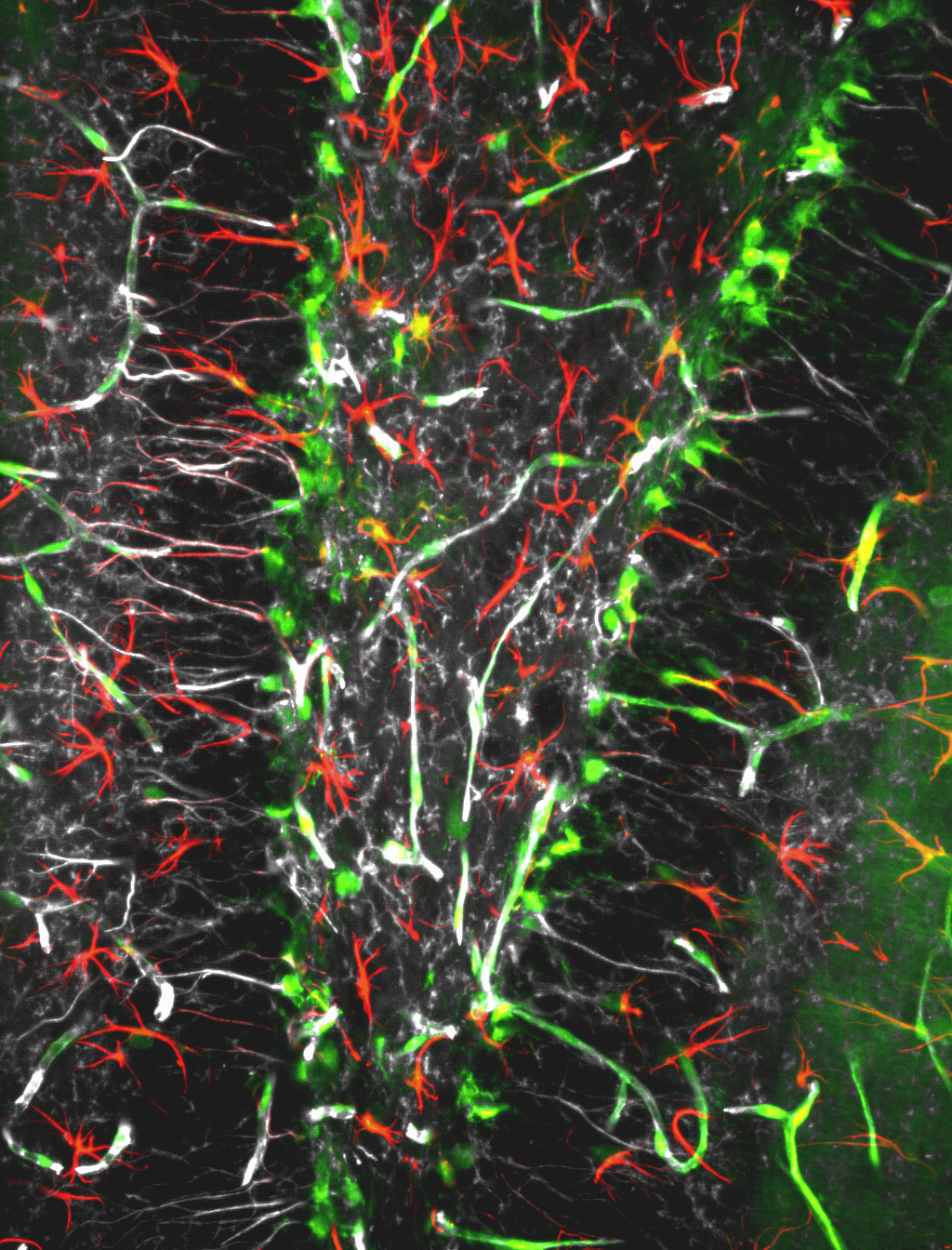
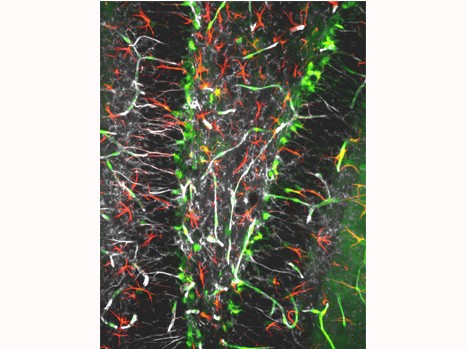 ©Inserm, Jinah Han. Among the hippocampal neural stem cells (characterised by expression of Nestin [in white] and GFAP [in red]), those that possess receptor VEGFR3 (in green) are very numerous.
©Inserm, Jinah Han. Among the hippocampal neural stem cells (characterised by expression of Nestin [in white] and GFAP [in red]), those that possess receptor VEGFR3 (in green) are very numerous.
