12 patients suffering from severe erectile dysfunction following prostate cancer received a transplant of stem cells into the penis. After six months, significant improvements in the quality of sexual intercourse, erection, penile rigidity and quality of orgasm were reported by the patients, according to a study by Inserm. This clinical trial was conducted under the direction of Professor René Yiou at the Department of Urology, Henri Mondor University Hospital, Paris Public Hospitals (AP-HP), Créteil.
These results are published in the journal European Urology.
Erectile dysfunction (sexual impotence) remains a common sequela of surgical removal of prostate cancer (radical prostatectomy). It is likely to severely affect the quality of life and self-image of men. Erectile dysfunction is caused by lesions in the vessels and nerves of the penis, which normally run alongside the prostate before reaching the erectile elements.
A pilot clinical trial was conducted by Inserm to repair the cellular lesions in the penis caused by radical prostatectomy. This new therapeutic approach consisted of injecting the penis with stem cells taken from the patients’ bone marrow. Many studies have shown that the bone marrow contains several types of stem cells that can spontaneously transform into cells of the same type as those damaged in the penis following radical prostatectomy (essentially endothelial, smooth muscle and connective tissue cells). They can also secrete substances that promote the repair of damaged blood vessels and nerves. In 2004, Prof. René Yiou’s team (Inserm Unit 955, “Mondor Institute for Biomedical Research”) at Henri Mondor University Hospital (AP-HP) set out to develop new therapeutic strategies to correct the sequelae of prostate surgery, using the regenerative capabilities of stem cells.
The main objective of this phase I/II clinical trial, supported by Inserm, was to test the feasibility and tolerance of an injection of bone marrow stem cells into the penis to treat erectile dysfunction following radical prostatectomy. The secondary objectives of the study were to evaluate the effects of the cell transplant on the quality of erections, sexual intercourse and the penile vessels. Since this treatment had not yet been tested in humans, it was necessary to conduct a first trial on a small number of patients to test patient tolerance to different doses of cells. For this pilot study, patients enrolled had severe erectile dysfunction considered to be irreversible following radical prostatectomy, i.e. the maximum medical treatment (prostaglandin injections into the penis + maximum doses of Viagra® and the use of a vacuum (erection pump[1])) remained ineffective after an average period of two years. Moreover, penile Doppler ultrasound showed that the erectile dysfunction was caused by severe damage to the penile vessels.
Trial methodology
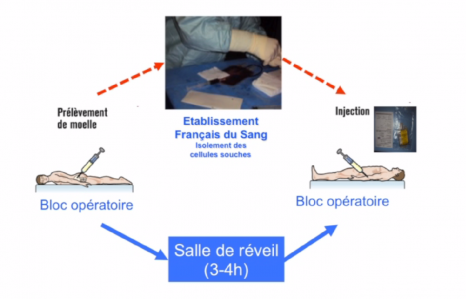
Twelve patients were enrolled for a total duration of 6 months. Four increasing doses of stem cells were tested, with each patient participating in the clinical trial receiving only one injection. The stem cells were taken from the bone marrow in the hip, and prepared by the French National Blood Service (Dr Hélène Rouard).
The effects of cell transplantation were assessed by specific self-administered questionnaires that scored the main aspects of sex life, namely satisfaction with intercourse, erectile and orgasm functions, libido, and penile rigidity during intercourse. The penile vessels were studied by Doppler ultrasound before and after cell transplantation. The researchers also assessed changes in the size of the penis after transplantation, since penile shrinkage is a common sequela of radical prostatectomy.

(c) Inserm / AP-HP
Main results
Tolerance of the treatment was excellent, and the main side-effect reported was transient pain at the puncture site where the bone marrow was taken (buttock region).
Six months after cell transplantation, the researchers noted a significant improvement in the main sexual scores, particularly a 10-point increase in the mean score for erectile function (17.4/30 at 6 months versus 7.3/30 before transplantation) on a scale of 0 to 30 (where 30 corresponded to the best possible erectile function). The other scores that improved significantly concerned overall satisfaction with sexual intercourse: 6.8/10 at 6 months versus 3.9/10 before transplantation (with 10 being the best score); the quality of orgasm: 6.3/10 at 6 months versus 3.5/10 before transplantation; penile rigidity during intercourse: 2.6/4 at 6 months versus 1.3/4 before transplantation (4 indicating maximum rigidity). At least two patients described a return to normal erections similar to the situation prior to radical prostatectomy, without taking drugs. A mean increase of 1 cm in penis length was observed. The clinical benefits were more pronounced at higher doses, and were associated with a normalisation of arterial vascular parameters in the penis measured by Doppler ultrasound. Improvement in sexual scores was maintained 1 year after transplantation, although some patients continued to use a treatment during intercourse.
In light of these good results, the researchers nonetheless wish to recall that: “Because of the small number of patients enrolled in the study, and the absence of a control group that would have received a ‘placebo’ injection, there is a need for caution about the conclusiveness of the efficacy shown for this new therapeutic strategy.” However, it is important to remember that the patients treated all showed severe erectile dysfunction that was unresponsive to the maximum medical treatment, and was associated with substantial damage to the penile vascularisation. The likelihood of spontaneous improvement in erections had been considered close to zero with these criteria. Improvement in sexual scores combined with a normalisation of Doppler ultrasound measurements of the penis argues in favour of a beneficial effect from the stem cell transplant in the medium term.
This therapeutic strategy could constitute the first approach to curing erectile dysfunction. For Prof. René Yiou: “If the results of this study are confirmed by other controlled clinical trials, the indications for cell therapy could be extended to other forms of erectile dysfunction that are less severe, or that result from common diseases such as diabetes or other vascular diseases.”
More information at: www.urologie-fonctionnelle.com
[1] The pump creates a vacuum around the penis, causing an influx of blood into the corpora cavernosa, and hence an erection.