We remember, right down to the tiniest detail, a car accident that happened over two years ago, when we feared for our lives; and yet, who among us can still remember a very good meal enjoyed just last year, even if it was really very, very good? Today, there is ever-increasing understanding of the biological bases behind this “adaptive” capacity to remember a stressful. However, until now, we knew very little about the state of post-traumatic stress disorder (PTSD). This pathological state is triggered in some individuals after exposure to a highly stressful event. In this state, patients are gripped by fear even when faced with elements that have no objective danger. Is PTSD specific to the human race, influenced by its history and culture? Or is it a state that is found in various species, resulting in common biological changes? These are the questions that Pier-Vincenzo Piazza, Director of the Neurocentre Magendie in Bordeaux (Inserm/ Université Victor Segalen), and his collaborators, have attempted to answer. Details of their results are provided in the Science review (advance publication on-line on Science Express website on 23 February).
It is easier to memorize a stressful event than an agreeable one. Almost all species capable of behaviour are able to recall negative events, proof that this is likely to be a capacity selected during evolution: it ensures survival in a hostile environment.
However, for some individuals, exposure to very stressful events may trigger a pathological state: post-traumatic stress disorder (PTSD) being the most emblematic. In the United States, estimates suggest that this syndrome afflicts 6.8% of the general population and that 30% of veterans from the Vietnam war and 12% of the Gulf war suffer from it (source National Centre for PTSD).
In this state of stress, the memory of the patient is impaired: it is no longer capable of adapting its reaction of fear to the “right” context and to the “right” predictive elements. Sufferers experience fear in situations where there are no threats. These fears become all-consuming, eventually preventing a normal life. “If you are attacked by a lion in the savannah as a flock of birds flew overhead, it would be normal to feel fear if you were to make a return trip to the savannah” explains Pier-Vincenzo Piazza. “However, you should not be gripped by fear if, when strolling around another natural open space, for example a golf green, you hear a bird cry or catch sight of birds on the horizon,” thethe Inserm research director goes on. If this is the case, you may have developed post-traumatic stress disorder as a consequence to the lion attack.
The teams, led by Pier-Vincenzo Piazza and Aline Desmedt, have demonstrated that such memory impairment associated with PTSD is not specific to humans and is also found in mice. To this end, the researchers conditioned mice to a) anticipate a threat (an electric shock of varying intensity) by using the same specific context (an indicating environment) and b) to distinguish this specific context from stimuli, which although present during the conditioning process, did not predict the threat (a sound).
In normal conditions, the mice showed a reaction of fear when exposed to the specific context (the indicator environment) of the threat, but did not react to the sound (not predictive of threat).
Following this conditioning session, the researchers then administered increasing concentrations of glucocorticoid hormones, the main biological response to stress in mammals. When administration of the glucocorticoids followed an intense threat, as is the case with individuals suffering from post-traumatic stress, the mice were no longer able to restrict their fear reaction to the “right” context and to the correct indicators pronouncing a possible threat. The animals began to show fear: they froze in response to indicators, which although present during the stressful situation, were not predictive of the threat in any way. These results show that PTSD is probably due to a simultaneous overproduction of glucocorticoids in some subjects as the traumatic event occurs.
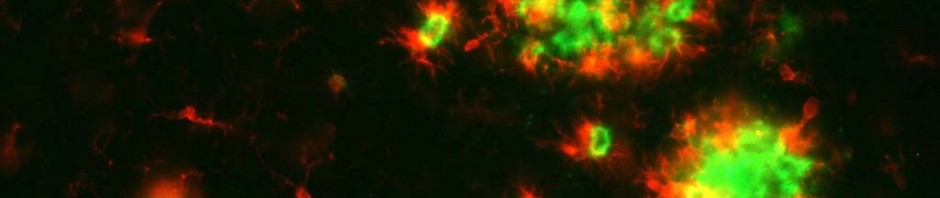
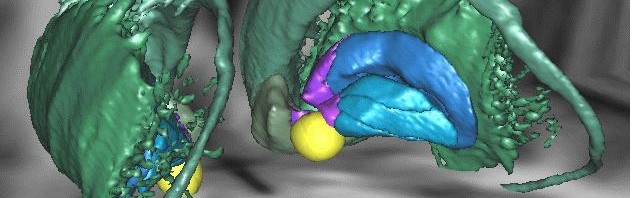
Memory impairment induced by glucocorticoids is accompanied by activity reorganisation in the brain, and, more specifically, the hippocampus-amygdale circuit, which is essential for coding memories associated with fear. In normal conditions, when an individual associates a threat with a context, strong activity is observed in the hippocampus, the structure in the brain required for all knowledge acquisition associating a specific context, area, etc. with an event. However, activity in the amygdale is low. The amygdale is an area of the brain that is also involved in emotional memory, but it memorizes specific indicators, such as sounds, which predict the threat.
When the mice were subject to an increase in glucocorticoids and memory impairment characterising PTSD was observed, activity in the hippocampus was reduced, whereas activity in the amygdale increased. In states of post traumatic stress, the researchers noted an inversion of normal activity in the brain. Normal activity in the amygdale may explain the fact that the subject begins to “over-respond” to presumed indicators, present during the traumatising event, but which are not, themselves, predictive of any danger. Low activity in the hippocampus may explain why subjects no longer recognize the right context: they are therefore incapable of containing their fear to appropriate situations.
“PTSD is not only an overbearing memory of the traumatizing situation, but also a memory impairment that prevents patients from containing their reaction of fear to the context that predicts the threat”, explain the researchers. In the case of post-traumatic stress disorder, a vivid memory of the traumatising event is associated with amnesia in terms of the surrounding context of the event. Some contextual elements present during the traumatising event are wrongly considered to be predictive of the event.
To conclude, the authors explain that PTSD-related memory problems seem to be caused by a biological response to abnormal stress suffered by some patients: for these patients, excessive glucocorticoid production at the same time as exposure to intense stress triggers an inversion of normal activity in brain structures that code fear-related memories.
“We have demonstrated that PTSD can occur in species other than humans and that there are shared biological origins. The mice model of this pathology now paves the way for better understanding of the molecular bases of this pathology, which could lead to the development of a treatment,” conclude the Inserm researchers.