Sorry, this article is only available in French.
Month: November 2012
The radical restructuring of brain networks in comatose patients
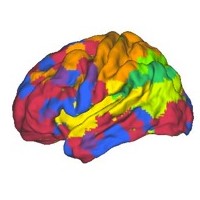
Researchers from Inserm, CNRS and the Université Joseph Fourier in Grenoble, in collaboration with Cambridge university, Strasbourg university and clinical practitioners from the Strasbourg University Hospital Centre, have analysed data from 17 comatose patients using functional MRI data. Their research reveals that the brain networks of these patients have been restructured. The results, published in PNAS on 26 November 2012, could help clinical practitioners diagnose comatose patients.
The researchers are focusing on analysing brain networks of brain-damaged comatose (non-traumatised) patients, a state where the individual is considered to be unconscious.
The authors of the study used an original graph theory-based methodology, where images were constructed using functional MRI data at rest and using robust statistical signal-processing methods. Local and overall effectiveness indices of functional brain networks were obtained for 17 brain-damaged patients and 20 healthy volunteers. Correlations in 417 brain regions were extracted to produce brain connection graphs using the statistically significant correlations.
Inserm unit 836 “Grenoble Institut des neurosciences”, CNRS researchers from the “GIPSA lab” and from the Behavioural and Clinical Neuroscience Institute in Cambridge, in collaboration with clinical practitioners from the Strasbourg University Hospital Centre, have been able to highlight restructured brain networks in brain-damaged (non-traumatized) comatose patients.
Through comparisons with the healthy subjects, the results demonstrate that the overall cerebral connectivity is preserved in comatose patients. By analysing the connectivity at a local level, the authors of the study have observed that some brain regions (“hubs”), which are highly connected in healthy volunteers, are less well connected in comatose patients. Conversely, the less densely connected regions in the network in healthy subjects become “hubs” in comatose patients.
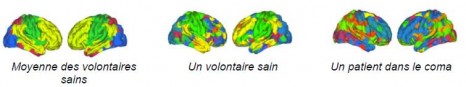
Brain imaging obtained from connectivity graphs
Photos: © Sophie Achard – Petra Vertes
The connectivity graph method is used to summarize in a single image data acquired through MRI scanning. It translates the effectiveness of connections in a single region compared to all the others. By grouping the most interconnected regions, modules are revealed (each represented by a different colour). Patients and healthy volunteers both have different models in their spatial location, representing radical alterations to the brain connections.
According to current hypotheses, consciousness disorders in persistently comatose patients could be linked to disconnection phenomena between specific cortical regions, particularly the precuneus. The results of this study also point in this direction. “From an overall perspective, the topology of brain connections resists well to traumatism by reorganising the most interconnected regions in the network. It therefore seems that comas may be linked to changes in the location of “hubs” among the brain networks” suggests Chantal Delon Martin, an Inserm researcher.
An assessment of brain injury and comas
Patients with brain injury may go through various clinically-defined states: vegetative state that is characterized by the preserved sleep-wake cycle (eyes opening spontaneously, autonomous breathing, etc.); minimally conscious state where patients have partially preserved environmental consciousness (eye movement capacity, reaction to stimulation); locked in syndrome where the patient is paralysed but conscious (communication using eyes); brain death when the coma is irreversible flat line EEG, no blood flow).
Coma (from the Greek κῶμα kôma meaning “deep sleep”) is one of the different states where self awareness and consciousness of the outside world is eradicated further to an accident (cerebral, cardiac, etc.). There are two coma phases: the “acute” coma phase (a few days after the accident) and the “chronic” phase (one month or more). Brain restructuring was observed by researchers during the “acute” phase, when it is not known which coma type the patient will develop.
Assessments of brain injuries in comatose patients are currently conducted through clinical examination, morphological MRI, evoked potentials and by SPECT (Single-photon emission computed tomography) or TEP (Positron emission tomography (PET). “The results of this study could help clinical practitioners in the difficult diagnosis process for comatose patients, since this method makes it possible to characterize each patient individually”, conclude the researchers.
Intestinal bacteria an aggravating factor in obesity-related illnesses
Researchers from INRA and Inserm have discovered that intestinal bacteria (“microbiota”) cause metabolic complications in the liver related to obesity. This is the first time such a link has been established using intestinal bacteria transplanted in mice. The results, published online in the journal Gut on 29 November 2012, pave the way towards preventive and therapeutic treatments based on controlling intestinal microbiota to cure type-2 diabetes and obesity-related liver conditions.
© Fotolia
Certain types of liver disease are linked to a condition called metabolic syndrome and develop in obese individuals in particular. The severity of these illnesses varies, from steatosis, in which lipids accumulate abnormally in the liver (a condition present in 80% of obese patients), to cirrhosis (present in 20 to 30% of all cases).
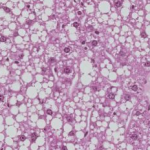
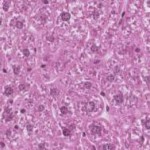
No two metabolisms react in exactly the same way to the same high-fat diet. Based on that premise, the INRA team, working with a team from Inserm, decided to study the role played by the billions of bacteria present in the digestive tract – known as intestinal microbiota – in the appearance or worsening of obesity-related liver diseases. To establish the precise effects of the bacteria, researchers transplanted microbiota into mice initially devoid of them. One group received intestinal microbiota from a mouse suffering from insulin resistance and steatosis, while another group received bacteria from a healthy mouse. Fed an excessively fat-rich diet for 16 weeks, all the mice became obese. Those which had been transplanted with microbiota from the insulin resistant and steatosis-afflicted mice, however, developed hyperglycaemia and hyperinsulinaemia, while the second group did not. Analysis of the liver tissue also revealed a more severe degree of steatosis in the first group, as confirmed by a higher concentration of lipids in the livers of these mice (see photos, below). Sequencing of the composition of the microbiota revealed the presence of different bacteria species in the two groups.
These results show that the composition of intestinal microbiota (determined by a wide variety of factors, including diet, genetic predisposition, and environment) plays a role in the development of metabolic syndrome-related illnesses. While the exact mechanisms of these bacteria remain unknown, the study suggests that controlling or transplanting intestinal microbiota could be a new way to prevent and/or treat type-2 diabetes and obesity-related liver disease.
Sections of liver in mice transplanted with microbiota from other mice presenting metabolic disorders (left) and with microbiota from healthy mice (right).
Only the mice transplanted with “bad” microbiota (left) developed severe steatosis when exposed to an excessively fat-rich diet (as evidenced by the clearly visible white fat droplets).
© Stephan Bouet, Histiology Workshop, GABI, INRA
(1) Metabolic syndrome is a medical condition known to precede the development of several serious illnesses, including type-2 diabetes, heart disease and stroke.
(2) Insulin resistance is typically present in cases of obesity. During periods of insulin resistance, the pancreas of an obese person secretes insulin which no longer has an effect on its receptors, a phenomenon known as desensitisation. Despite the presence of insulin, glucose enters cells less effectively and accumulates in the bloodstream, increasing blood sugar levels (hyperglycaemia). This in turn provokes a hypersecretion of insulin (hyperinsulinaemia).
November 29th 2012
Sorry, this article is only available in French.
November 28th 2012
Sorry, this article is only available in French.
November 27th 2012
Sorry, this article is only available in French.
November 24th 2012
Sorry, this article is only available in French.
November 23th 2012
Sorry, this article is only available in French.
Predicting the age at menopause of women having suffered from childhood cancers

Researchers from Inserm, the French public hospital organization (AP-HP), the Institut Gustave Roussy and the Université Paris-Sud studied the age at menopause of a cohort of 706 women who had suffered from childhood cancer. The data revealed that these women, in particular those who had undergone unilateral oophorectomy or who had been treated with alkylating agents and who had received a radiation dose to the ovaries, were menopaused on average 4 to 7 years earlier than average. This study provided important data about the fertility window of women who had suffered from childhood cancer and information concerning the associated risk factors, but did not confirm the greater risk of premature menopause (before the age of 40) that was reported by the American studies.
The results were published in the review Human Reproduction of November 15.
Women who have suffered from childhood cancer are known to run a greater risk of premature menopause. However, data about the associated risk factors is limited. Researchers from unit 1018 “Centre for Research in Epidemiology and Population Health (CESP)” (Inserm/Université Paris-Sud/Institut Gustave Roussy) and from the AP-HP analyzed the data from a French cohort, named Euro2k, concerning 1522 survivors of childhood cancer diagnosed between 1945 and 1986 when they were under 18, initially in order to study the mortality rate. The study estimated the radiation doses received at the ovaries by the women in this cohort who had been treated by radiotherapy. 706 of these women filled in a detailed questionnaire about their state of health. 32% of these women had already reached the age of 40 years; 7% were over 50 years of age. The research team studied the age at menopause of these women and the potentially associated risk factors. The researchers based this study on self-reported questionnaires sent to the women in order to obtain information about the menopause, without confirming by measuring FSH levels.
Analysis of this data showed that 97 women (13,7%) were menopaused at a median age of 44 years, in other words, 7 years earlier than the general population. For a third of these women (36%), menopause was surgically induced.
The researchers concentrated on the risk factors of menopause in these women, who had been subjected to various cancer treatments during childhood. It appeared that being treated during puberty was associated with a risk of non-surgical menopause. At a given age, the maximum risk of early menopause was observed in women who had been treated after the onset of puberty with alkylating agents (either alone or along with even a minimum dose of radiation to the ovaries, for example 0.01 Gray). Menopause occurred on average 4 years earlier in women who had been exposed to these agents. Having undergone unilateral oophorectomy is also associated with a 7-year earlier age at menopause.
The results showed that women who had suffered from a childhood cancer were more likely to suffer from early menopause, but did not conform the high risk of premature menopause (in other words, before 40 years of age), such as was reported in the American studies. This is probably due to the difference between the populations studied (there were no cases of leukemia or lymphoma in the Euro2k cohort population).
“In this cohort , very few women had received high doses of radiotherapy in order to receive bone marrow transplants, and only 21% of them (i.e. twice the occurrence of the general population) suffered premature menopause before the age of 40”, explained Cécile Teinturier, the principal writer of the study. The main risk factors associated to these cases of premature menopause are: the older the women is when being treated for cancer, the dose of alkyating agents such as Cyclophosphamide or Melphalan received during bone marrow transplants, and the radiation dose received at the ovaries.
“This study provided information about the risk factors affecting the fertility window of women who have suffered from childhood cancer. This new data should help both to inform patients who are at risk of premature menopause, by advising them not to delay their first pregnancy until after the age of thirty, and to reassure women who present a low risk” concluded Cécile Teinturier.
It is planned to extend Cohort Euro2k to cover all patients treated for solid cancers who were under 18 years of age before the year 2000 inFrance. The aim is to study the impact of high-dose chemotherapy on the occurrence of premature menopause.
November 22th 2012
Sorry, this article is only available in French.