Researchers at Inserm Unit 1194, “Montpellier Cancer Research Institute” (Inserm/University of Montpellier/Montpellier Regional Cancer Institute) have confirmed the value of a new test to identify cancer patients who will be free of sequelae following radiotherapy. This test, conducted on a blood sample taken from 500 breast cancer patients, treated in 10 centres in France, and monitored for 3 years, showed that women with a high rate of radiation-induced lymphocyte apoptosis (RILA) had a very low rate of late breast fibrosis. These results, which are published in EBioMedicine, suggest that personalisation of curative intent radiotherapy could be considered, with tailoring of the radiation dose delivered to the patient and the radiotherapy technique employed.
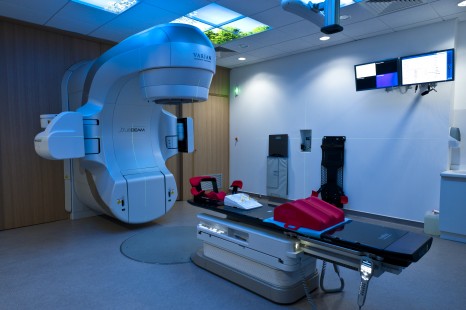
Radiotherapy is one of the treatments used to treat breast cancer. The irradiation destroys the malignant cells in a targeted manner. However, it also leads to the death of some healthy cells in the irradiation field. Using a single blood sample, the researchers analysed the rate of radiation-induced CD8 lymphocyte apoptosis (RILA) in the context of a prospective multicentre clinical trial which began in 2005. The objective of this trial is to develop a functional test based on RILA to predict radiosensitivity in tissues. The trial follows several pilot trials initiated in the last 15 years in relation to breast cancer, as well as other diseases.
In this context, 500 female patients with breast cancer and treated using radiotherapy were recruited in 10 French centres. The researchers at the Joint Research Unit “Montpellier Cancer Research Institute” (Inserm/University of Montpellier/Montpellier Regional Cancer Institute) performed RILA at 8 Gy for the patients before they underwent radiotherapy. The patients were then monitored for three years in order to assess late breast sequelae (fibrosis).
Results of the multicentre study provide large-scale confirmation of the preliminary data obtained by the researchers. They show that a high RILA value is correlated with a low incidence of late sequelae. A low rate of late breast fibrosis was observed, with a negative predictive value of over 90%. Conversely, almost all patients who showed a high level of fibrosis corresponded to the group with a low RILA value, predictive of more pronounced sequelae.
“This multicentre study provides a sufficient level of proof to allow the use of this test in routine clinical practice, and changes patient management. Given the results obtained, we can consider the possibility of increasing the total irradiation dose delivered locally, or of modifying the target volumes without compromising the oncological outcome,” explains David Azria, principal investigator in the study.
In practice, this test is carried out by taking a single blood sample, and results are obtained in 72 hours.
By providing the opportunity to identify patients who are not prone to sequelae and those at greatest risk, this test paves the way for the personalisation of curative intent radiotherapy.
It should not be used alone, but should be combined with other parameters in a predictive nomogram, a useful calculation tool, for which a patent application has been filed by the Montpellier team. “The results, when combined with the all the parameters, provide a reliable estimate of the risk of late sequelae following radiotherapy,” concludes David Azria.