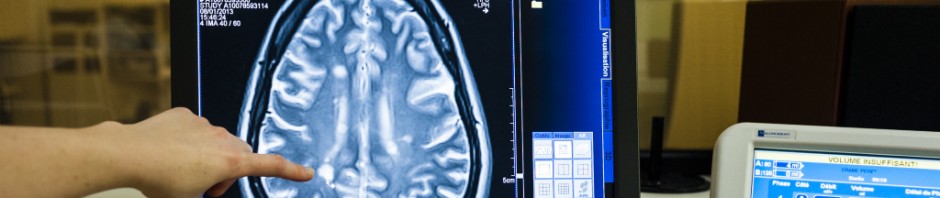
Under normal conditions and because it cannot store oxygen, the brain cannot withstand being deprived of oxygen for more than a few minutes without risking serious consequences. After an accident (cranial trauma or stroke), emergency teams therefore try to restore cerebral oxygenation as quickly as possible. The faster and more precisely physicians work, the greater the chances of recovery. A multi-disciplinary team at the Grenoble Institute of Neuroscience (GIN, Inserm/ Grenoble-Alps University/Grenoble teaching hospital) comprising physicists, biologists and physicians (neurologists and anaesthetists) has developed a new method for measuring cerebral oxygenation using MRI. Besides being non-invasive, this technique identifies the least oxygenated areas of the brain with precision. Ultimately, it could be used to guide therapeutic interventions and make them more precise, less risky and more effective.
These results are published in the Journal of Cerebral Blood Flow and Metabolism.
Although the brain accounts for just 2% of bodyweight, it uses 20% of the total oxygen consumed by the body. It is unable to create ‘reserves’ and the slightest shortage of oxygen can result in serious consequences (e.g. loss of language functions and motor skills) and even death if not restored very quickly. Ideally in a trauma situation, it should be possible to immediately identify the areas of the brain most affected by a lack of oxygen and where possible avoid physically operating on already weakened bodies.
After an accident, it is vital to monitor oxygenation of the brain. Nowadays, standardized international methods, notably the installation of a probe in the brain, allow brain oxygenation to be estimated locally. This operation entails a neurosurgical procedure and only allows a highly localized measurement of cerebral oxygenation. The only means of mapping the brain’s oxygen levels is by a radioactive measurement of oxygen using functional imaging (a technique developed in the 80s). However, this method also has its limits as it is expensive and requires PET (positron emission tomography) equipment which is scarce in France. Finally, while the brain’s total oxygen consumption remains constant, it varies between the various regions of the brain.
The new MRI method developed by Inserm researchers measures brain tissue oxygen saturation or the quantity of oxygen present in the tissue microvascularization (expressed as StO2).
Thanks to this technique, researchers have observed a major reduction in cerebral oxygenation in an animal model ischemic stroke and, to a lesser extent, following cranial trauma. By mapping cerebral oxygenation, it has been possible to identify regional heterogeneity of StO2 in strokes, cranial trauma and a model brain tumour.
This innovative approach offers a number of benefits including the possibility of immediate use with humans. Unlike positron emission imaging (or PET imaging), which is the gold standard technique, no radioactive tracer needs to be injected when performing an MRI examination. StO2 maps with enhanced spatial resolution can also be produced by MRI. Moreover, MRI examinations are less expensive than PET examinations and MRI imaging devices are more widespread in France than PET imaging devices.
The results of this preclinical trial match those achieved with the two gold standard methods (measurement of blood gas levels and histological mapping of oxygen deprived areas) for different oxygenation levels.

From a clinical perspective, several applications may be foreseen, which are illustrated in this project by 3 situations:
- In patients suffering from cranial trauma, this map could help identify the least oxygenated areas of the brain. Follow-up and therapeutic strategy could therefore be adapted to optimal effect.
- In stroke patients, this technique should enable more effective identification of the area of ischemic brain tissue which is likely to recover after treatment.
- Finally, certain malignant brain tumours contain poorly oxygenated areas which are known to be resistant to treatments such as chemotherapy and radiotherapy. It is strongly suspected that these poorly oxygenated areas contain tumour stem cells which cause tumour recurrence. By mapping them more effectively, it should be possible to adapt and customize treatments.
Although the reliability of this system has now been established, it remains to be seen to what extent it is capable of guiding the work of neurologists and anaesthetists. “In several years’ time, it is possible that a precise map of brain oxygenation will enable us to supply drugs to the right place or more effectively configure surgical operations to reduce intracranial hypertension” explains Emmanuel Barbier, Inserm research director.
This work was carried out using the IRMaGe platform, which is a platform of the ‘France Life Imaging’ national life imaging infrastructure. It was funded notably by the Agence Nationale pour la Recherche [French National Research Agency] (IMOXY project).